Compression neuropathies of the radial nerve
Mark A. Pickford 2
1 Plastic and Hand Surgery, Hinchingbrooke Hospital, United Kingdom
2 Queen Victoria Hospital, East Grinstead, United Kingdom
Abstract
The commonest cause of radial nerve dysfunction is from extrinsic compressions of the radial nerve, such as against the shaft of the humerus in “Saturday night palsy”. However, this chapter focuses on the less common intrinsic compression neuropathies of the radial nerve. Intrinsic radial nerve compression neuropathies occur much less frequently than those of the median or ulnar nerves. The combined annual incidence of compressive neuropathies of the posterior interosseous nerve and the superficial radial nerve is only 0.003%, compared to an annual incidence 0.1–0.35% for carpal tunnel syndrome and 0.03% for ulnar nerve compression syndromes. Three radial nerve compression syndromes are described: Radial Tunnel Syndrome (RTS); Posterior Interosseous Nerve (PIN) syndrome; and compression of the Superficial Radial Nerve (SRN) in Wartenberg’s syndrome [1].
Radial nerve anatomy
Knowledge of radial nerve anatomy is required in understanding and differentiating these three radial nerve compression syndromes. The posterior cord of the brachial plexus becomes the radial nerve once the axillary nerve branches off through the quadrangular space at the lower border of teres minor muscle. The radial nerve exits the axilla via the apex of the lateral muscular triangle to enter the posterior compartment of the arm. Lying deep to triceps, the radial nerve passes from supero-medial to infero-lateral through the spiral groove on the posterior aspect of the humerus. At approximately the junction of the middle and distal third of the arm the radial nerve turns anteriorly, around the lateral border of the humerus, and pierces the lateral antebrachial septum to enter the anterior compartment of the arm 10 to 12 cm above the elbow. Here it enters the radial tunnel, a musculofascial space formed in its proximal part by biceps and brachialis muscle antero-medially, brachioradialis, extensor carpii radialis longus and extensor carpii radialis brevis muscle antero-laterally; the radio-capitellar joint capsule forms the floor. Within the radial tunnel the radial nerve divides into the posterior interosseous nerve (PIN) and sensory branch of the radial nerve (SRN) 6.0 to 10.5 cm distal to the lateral intermuscular septum and 3 to 4 cm proximal to the aponeurotic leading edge of the superficial (humeral) head of the supinator muscle (the Arcade of Frohse). The SRN travels deep to brachioradialis muscle until around 8 cm proximal to the radial styloid, where it pierces the deep fascia at the lateral edge of brachioradialis muscle to run superficially and supply sensation to the radial distribution of the dorsum of the hand. The PIN passes in an oblique course between the two heads of supinator muscle to enter the posterior compartment of the forearm [2], [3]. The PIN supplies motor innervation to all the extensor muscles of the wrist and digits except for the extensor carpi radialis longus (ECRL) which is supplied by a branch typically from the radial nerve prior to its division into the PIN and SRN. The PIN divides into a medial (superficial) branch innervating extensor carpi ulnaris muscle (ECU), extensor digiti minimi muscle (EDM), and extensor digitorum communis muscle (EDC), and a lateral (deep) branch to abductor pollicis longus (AbPL), extensor pollicis brevis (EPB) and longus (EPL) and extensor indicis muscle (EI) [4]. The PIN is often described as a “motor-only” nerve, but it does also carry sensory afferent fibres from the wrist, as well as group A beta afferent nerve fibres from the muscles along its distribution [5].
Radial tunnel syndrome (RTS)
Introduction
Michele and Krueger first described radial tunnel syndrome in 1956. They used the term radial pronator syndrome, as they identified axial rotation of the forearm in the aetiology of this condition [6]. Since then radial tunnel syndrome has remained one of the major controversies in upper limb surgery. Does it even really exist, what is its incidence and how do you clinically test for it?
Pathophysiology
Anatomically there are five potential sites of compression of the radial nerve within the radial tunnel (see figure 1).
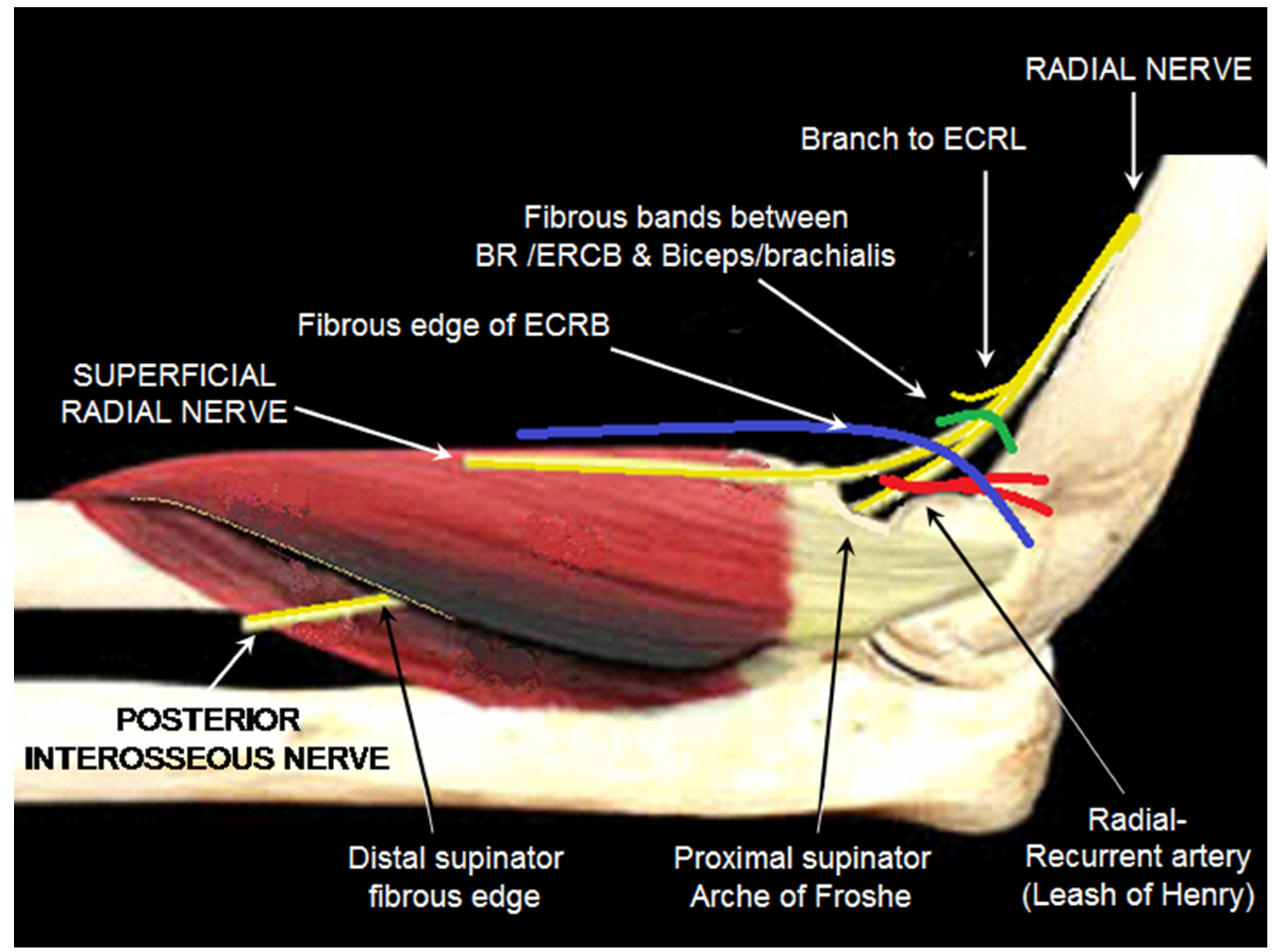
From proximal to distal these are:
- fibrous bands between the biceps/brachialis muscles medially and brachioradialis/ECRB muscles laterally [7];
- the fibrous edge of ECRB arising from the common extensor origin, which grooves the nerve when the forearm is in pronation [8];
- the “fan” of vessels arising from the radial-recurrent artery, the so-called “Leash of Henry” [9] which cross the nerve to supply the brachioradialis muscle, epicondylar region and elbow joint;
- the leading edge of the supinator muscle, referred to as the arcade of Frohse [10], [11];
- the distal edge, or even within, the supinator muscle (although many authors do not include this fifth section as part of the radial tunnel) [12], [13].
These final two sites only compress the PIN, which passes between the two heads of the supinator muscle. The most common site is at the proximal edge of the superficial head of supinator, with a tendinous arcade of Frohse found in 30% to 100% of anatomical studies [3], [5], [8], [14], [15]. Intraoperatively passive stretch of the supinator has been demonstrated to increase pressure on the PIN to 40–50 mmHg. This is sufficient to inhibit venous blood flow of vasa nervorum, as well as block axonal transport [16]. A thickened leading edge of the extensor carpi radialis brevis muscle is the second most common site of compression [14].
Clinical aspects
Patients with RTS complain of pain along the lateral proximal forearm, which may radiate down to the dorsum of the wrist, or from the wrist up to the elbow [17]. Radial tunnel syndrome (RTS) is a clinical diagnosis of exclusion, as there are no objective measurements in identifying this condition. The dominant arm is more frequently affected, and individuals performing repetitive activities of pronosupination, such as athletes and labourers, are at increased risk. Men and women are equally affected, and the peak incidence is in the 4th to 6th decades of life [13].
Approximately 5% of patients who initially present with so-called tennis elbow actually have radial tunnel syndrome, with such patients often being labeled as having “recurrent tennis elbow” [14]. RTS is differentiated from lateral epicondylitis as the pain is located distal to the lateral epicondyle, typically around the mobile wad area, and the pain increases with activity. Weakness and paralysis are not part of the clinical picture of RTS.
Provocative tests
Compression over the radial tunnel 3 to 4 cm distal to the lateral epicondyle, over the mobile wad area, should reproduce the pain. Loh and colleagues describe this as the “rule-of-9” test. They divide the volar forearm into a grid pattern of nine equal sized boxes, such as in the game “noughts and crosses”. Pain from pressure over the proximal and middle box in the lateral column of boxes corresponds with the course of the radial nerve through the radial tunnel and under the supinator muscle, and is regarded as diagnostic of radial tunnel syndrome [18], [19].
With the elbow extended, the forearm pronated and wrist flexed, maximal traction is put through the PIN and radial nerve, and should reproduce RTS pain [20]. Resisted supination or resisted wrist extension are also described in diagnosing RTS. Different authors describe the resisted supination test with the elbow either extended or flexed 90 degrees and held in next to the body [17]. In addition Lister’s resisted middle finger extension test [5] should reproduce pain over the lateral proximal forearm. Although no comparative study of clinical tests for RTS has been done, Lister’s test is described as one of the more popular and reliable tests. Resisted extension of the middle finger with the elbow extended should cause sharp pain in the region of the extensor origin as there is particularly strong contraction of the ECRB which inserts onto the middle finger metacarpal. The patient automatically pronates the forearm with this test such that the extensor wad, but in particular the fibrous edge of ECRB, impinges on the radial nerve/PIN in the radial tunnel [5], [7].
Diagnostics
Sarhadi et al. described injecting the point of maximal tenderness with local anaesthetic and steroid. If the positive provocative tests for RTS become negative after the injection of local anaesthetic then a diagnosis of RTS is made [17].
Electrophysiology tests
Most authors report no significant findings on imaging or electrodiagnostic studies with provocative tests regarded as the most reliable means of diagnosing RTS [20], [21].
However, Rosen and Werner [22] noted altered PIN motor latencies with active contraction of the supinator in 28 patients with radial tunnel syndrome. In addition, Kupfer et al. demonstrated how dynamic electrophysiological testing of PIN motor latencies may provide objective evidence of RTS. They found significant differences in radial nerve conduction during a provocative test maneuver when compared with “at rest”. These differences were considerably more significant in patients awaiting RTS surgery when compared with controls, and PIN motor latencies significantly improved following radial tunnel decompression in these same patients [21].
MRI
Ferdinand et al. retrospectively reviewed MRI scans of 25 patients with a clinical diagnosis of RTS to substantiate anecdotal evidence of increased signal intensity seen in PIN innervated muscles in RTS [23], [24]. Denervation oedema or atrophy within the supinator or forearm extensor muscles were seen in just over half the patients, and 25% had an identifiable cause of PIN/radial nerve compression:
- thickened leading edge of the extensor carpi radialis brevis (n=4)
- prominent radial recurrent vessels (n=1)
- schwannoma (n=1)
- bicipitoradial bursa (n=1)
In 2 patients lateral epicondylitis was identified. MRI images did not always correspond with the operative findings, and there is no mention of the electrophysiological study results, however it demonstrates that causes of mass effect within the radial tunnel may be identified through MRI.
Management
Surgery has been reported by many to give satisfactory results [5], [7], [14], [17], [25]. This supports the existence of RTS, and has been used as confirmation of the diagnosis in patients relieved through surgical decompression. However, there is no level 1 evidence, and many of the studies do not stand up to rigorous analysis. Roles and Maudsley include patients with posterior interosseous weakness or palsy in their 36 patients described as having RTS. Sarhadi et al. reviewed 26 patients with RTS, of which 9 underwent surgery. However, half had other confounding upper-limb pain syndromes (tennis elbow –8; carpal tunnel syndrome –6; intersection syndrome –1; cubital tunnel syndrome –1; De Quervains disease –2, thoracic outlet syndrome –1; trapeziometacarpal joint arthritis –2). Atroshi et al. also noted that operative findings do not always correspond with the symptoms and outcomes.
Controversy is fueled by the fact that the natural history of the condition is not known. High cure rates are reported by surgery on RTS, however Sarhadi et al. found that the majority of cases in their series responded to conservative treatment with physiotherapy or steroid injection (17/26), and 16 patients continued to have long term pain relief at 2 years. Only 9 out of 26 failed conservative measures and progressed to surgery. This argues strongly for surgery only being offered to those that fail to respond to conservative management.
Posterior interosseous nerve (PIN) syndrome
Introduction
The first report of an isolated PIN palsy was in 1905 in an orchestral conductor whose palsy was attributed to the repetitive pronosupination of conducting [29]. Occupations with repetitive pronosupination have since been considered a risk factor for PIN syndrome with case reports in athletes [30], musicians [31] and manual professions such as bartending [32].
Clinical examination and diagnostics
PIN syndrome is characterised by a pain-free inability to extend the digits of the hand, or to extend the wrist in a dorso-ulnar direction (Figure 2).
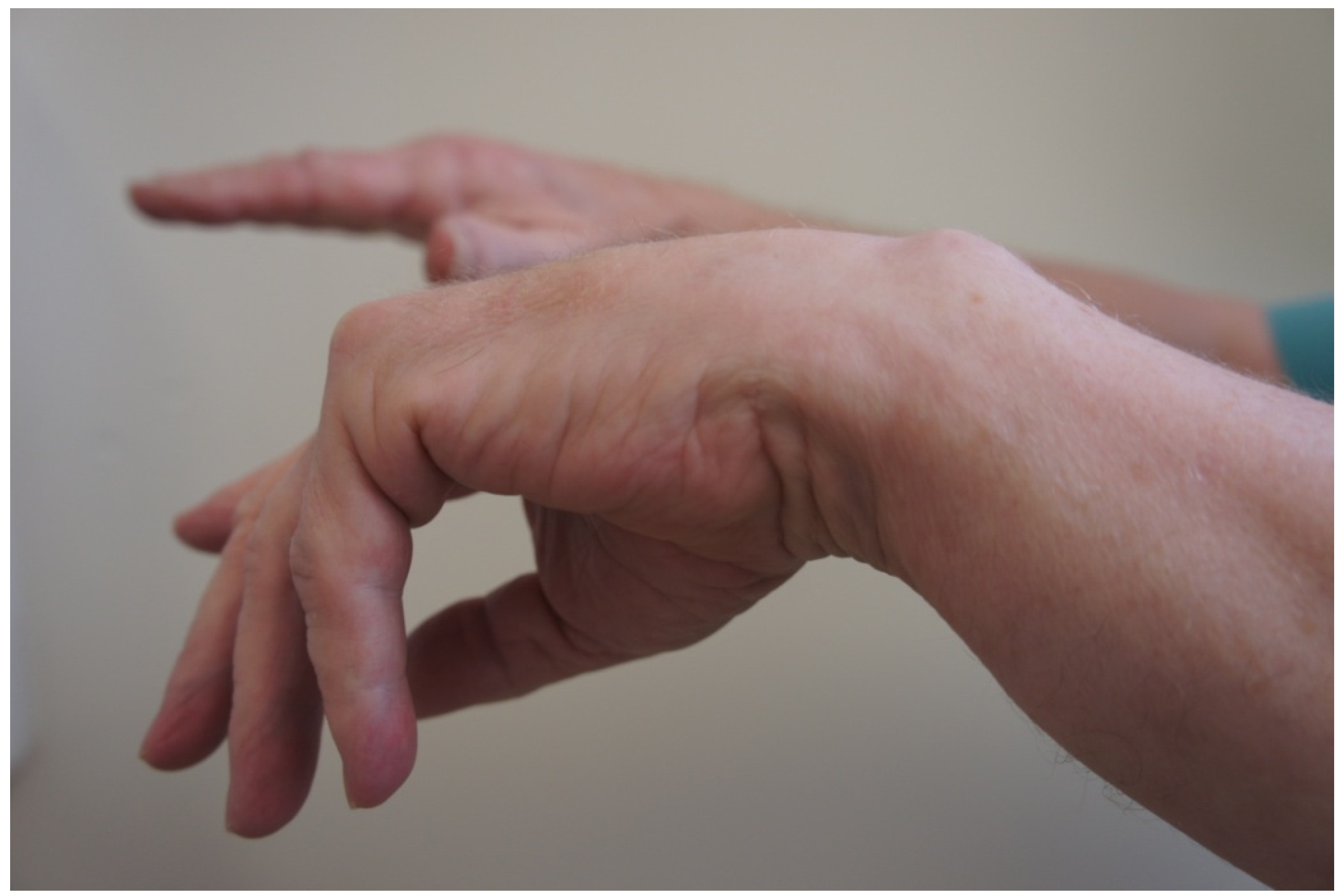
ECRL is spared in PIN syndrome as its motor branches arise from the radial nerve. This differentiates PIN syndrome from a more proximal lesion of the radial nerve in which wrist drop is seen. In PIN syndrome the wrist is held in radial deviation at rest, and dorso-radial extension is preserved. ECRB innervation can also arise directly from the radial nerve (in 71% of specimens according to one study [3]) such that the wrist may not radially deviate when extended. Sensation on the dorso-radial aspect of the hand is intact as the SRN is spared. Incomplete palsy may be seen in situations where only certain branches of the PIN are affected. For instance weakness of ECU, EDM, and EDC is seen with compression of the medial (superficial) PIN branch, whereas compression of the lateral (deep) branch causes weakness of the AbPL, EPB and EPL and EI [4].
Anatomy
The PIN enters the supinator muscle approximately 3.6 cm after its bifurcation from the radial nerve, and courses for just under 4 cm through the muscle (approximately 50% of the its length) [3], [15]. This short premuscular portion may be compressed by aetiologies more commonly associated with radial tunnel syndrome (fibrous bands at the level of the radial head, radial recurrent vessels (the Leash of Henry) and the tendinous margin of ECRB).
Both the proximal and distal ligamentous margins of the supinator are known sites of compression, however the PIN is most commonly compressed at the proximal margin, known as the Arcade of Frohse (AF) [33]. Spinner, in 1968, described the AF and its relationship to PIN palsy, reporting 2 distinct heads of the supinator muscle. He described the superficial head as tendinous in 30% of adult arms [8], with other investigators reporting a tendinous AF in 57% [15] to 100% of the arms examined [5]. Thomas et al. found that only 32% of specimens had a tendinous arcade, and that there was a clear association with the shape of the muscle. The supinator muscle had two heads (as described in anatomical texts) in only 29% of specimens, in which the superficial head from the lateral epicondyle always had a tendinous arcade [3]. 71% of specimens had a single “semi-circular” head, originating from the elbow joint capsule, through which the PIN passed. Only one had a tendinous arcade.
During its passage between the two heads of supinator the PIN divides into its medial/superficial branch (innervating EDC, EDM and ECU) and lateral/deep branch (innervating APL, EPB, EPL and EI) [8]. Three clinical pictures of PIN palsy are observed according to the site and specific branch of the PIN being compressed. Suematsu and Hirayama’s [34] describe these as:
- type 1 (drop fingers and thumb) – compression of the PIN as it enters the supinator prior to branching
- type 2 (drop fingers) – compression of the medial (superficial) branch of the PIN
- type 3 (drop thumb) – compression of the lateral (deep) branch of the PIN
In their study of 11 patients, all type 1 patients were tender to palpation over the proximal supinator, and were found to have compression of the PIN as it entered the supinator during surgical release. Type 2 and 3 patients were tender over the distal border of supinator and had compression of the medial and lateral PIN branches, respectively, by fascial thickenings at the exit point from the supinator muscle. They propose that preoperative tenderness should guide the surgeon to the point of compression and therefore the appropriate surgical approach. The PIN should then be exposed between the brachioradialis and brachialis muscle in type 1 patients, with the supinator muscle explored from its proximal border. In type 2 and 3 patients the PIN should be exposed between the ECRB and EDC and the supinator muscle explored from its distal border.
Owing to the relative infrequency of PIN syndrome no firm data exists on the frequency of the various aetiologies. Lipomas should be considered as a causative factor, as they are the most commonly reported tumour causing PIN syndrome [35], [36], [37], [38]. Ganglia are also commonly described, most frequently arising from the anterior capsule of the proximal radioulnar joint [39], [40], [41], [42]. Other reported sources of compression include vasculitis [43], rheumatoid pannus [44], and septic arthritis of the elbow [45].
The diagnosis of PIN syndrome is made from the history and clinical examination, with the characteristic finding of palsy of the finger and thumb extensors in the absence of trauma, tendon ruptures/subluxations or more proximal neurology. Tendon subluxations may result in an extension deficit at the metacarpophalangeal joints, however extension can be maintained after passive correction. The tenodesis effect, seen with moving the wrist from a position of extension to flexion, determines whether tendon ruptures have occurred.
Diagnostics
Electrodiagnostic studies should be used for additional confirmation and are usually positive (in contrast to radial tunnel syndrome) due to the loss of motor function. Electrodiagnostic studies also rule out more proximal causes of neurology, such as cervical spine, brachial plexus or proximal radial nerve lesions. MRI is useful as it may reveal space-occupying lesions or demonstrate secondary muscle oedema distal to the point of compression [34].
Unifying theory
Although PIN syndrome and RTS are defined as separate entities, the distinction between them is certainly not as clear as described in most texts. PIN syndrome is characterised by palsy, but in fact can often be heralded by pain. Similarly, RTS is characterised by pain and the absence of any palsy, however it can be associated with weakness on close examination. The presence of muscle denervation on MRI scans of patients with RTS suggests that RTS and posterior interosseous syndrome are parts of a continuum. Although predominately a motor nerve, the PIN does also carry sensory afferent fibres from the wrist and muscle mechanoreceptors [5]. The diameter of a nerve axon (motor being greater than sensory), the location of an axon within a nerve (peripheral vs. central), and whether the nerve is myelinated or unmyelinated, will determine their susceptibility to pressure effects, and may explain the differing presentations between radial tunnel and posterior interosseous syndromes [46].
Management of RTS and PIN syndrome
RTS and PIN syndrome should both initially be treated non-surgically with splintage, physiotherapy and anti-inflammatory medication, along with resting the arm. Activity modifications should be investigated, especially in RTS in order to eliminate provocative positioning of the arm. Splintage, to prevent joint contractures or shortening of the intrinsic and long flexor muscles, is the most important early intervention for PIN syndrome. Hand exercises should be performed to maintain a full passive range of motion of all joints [20].
MRI, or other appropriate imaging, should exclude a surgically correctable cause, such as a lipoma. Injection of steroid is accepted treatment for RTS and may also have a role in PIN syndrome. If there is no return in motor function at around 3 months spontaneous recovery is unlikely and therefore surgical exploration is recommended in PIN syndrome. Without intervention muscle fibrosis of PIN-innervated muscles occurs by around 18 months, by which time tendon transfer surgery is more appropriate than decompression surgery [20].
Surgery
Success from surgical decompression of RTS ranges between 67% [47] to 97% [7], with the success rates appearing to vary according to the number of patients with lateral epicondylitis, workers compensation or other nerve compression syndromes who were included in the studies. However, as RTS is a pain-only phenomenon, surgical success in alleviating pain has not been matched with patient satisfaction, which is reported at around only 40% in two studies [25], [48].
The argument for surgical decompression in PIN syndrome is stronger. Hashizume et al. [43] performed decompressions 2.2 months following PIN palsy in 17 patients, with 16 (94%) having full recovery at 4.5 months after surgery. In a smaller study of 8 patients Vrieling et al. [49] performed surgery at an average of 4.7 months post PIN palsy and showed a 75% good to excellent result at 5 years. This makes the case for earlier surgical intervention in cases of PIN syndrome.
Surgical approaches
Surgery for PIN and RTS is similar, with four standard approaches described; volar, lateral, dorsal and muscle splitting.
Volar approach (author’s preferred method)
A volar longitudinal incision between brachialis and brachioradialis muscles (extending from a few centimeters above the elbow joint to a quarter of the way down the forearm) is used for RTS (Figure 3a–b).
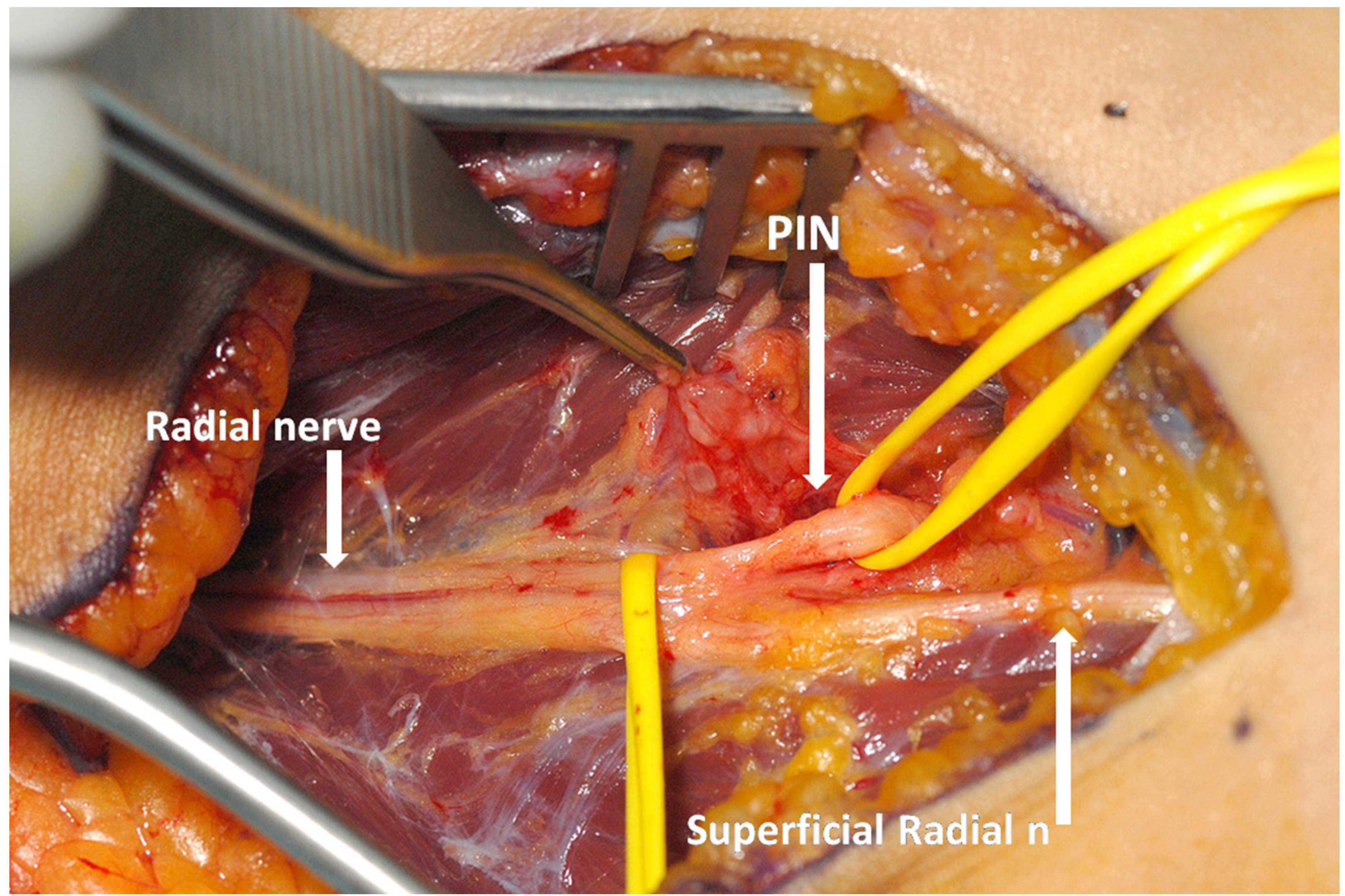
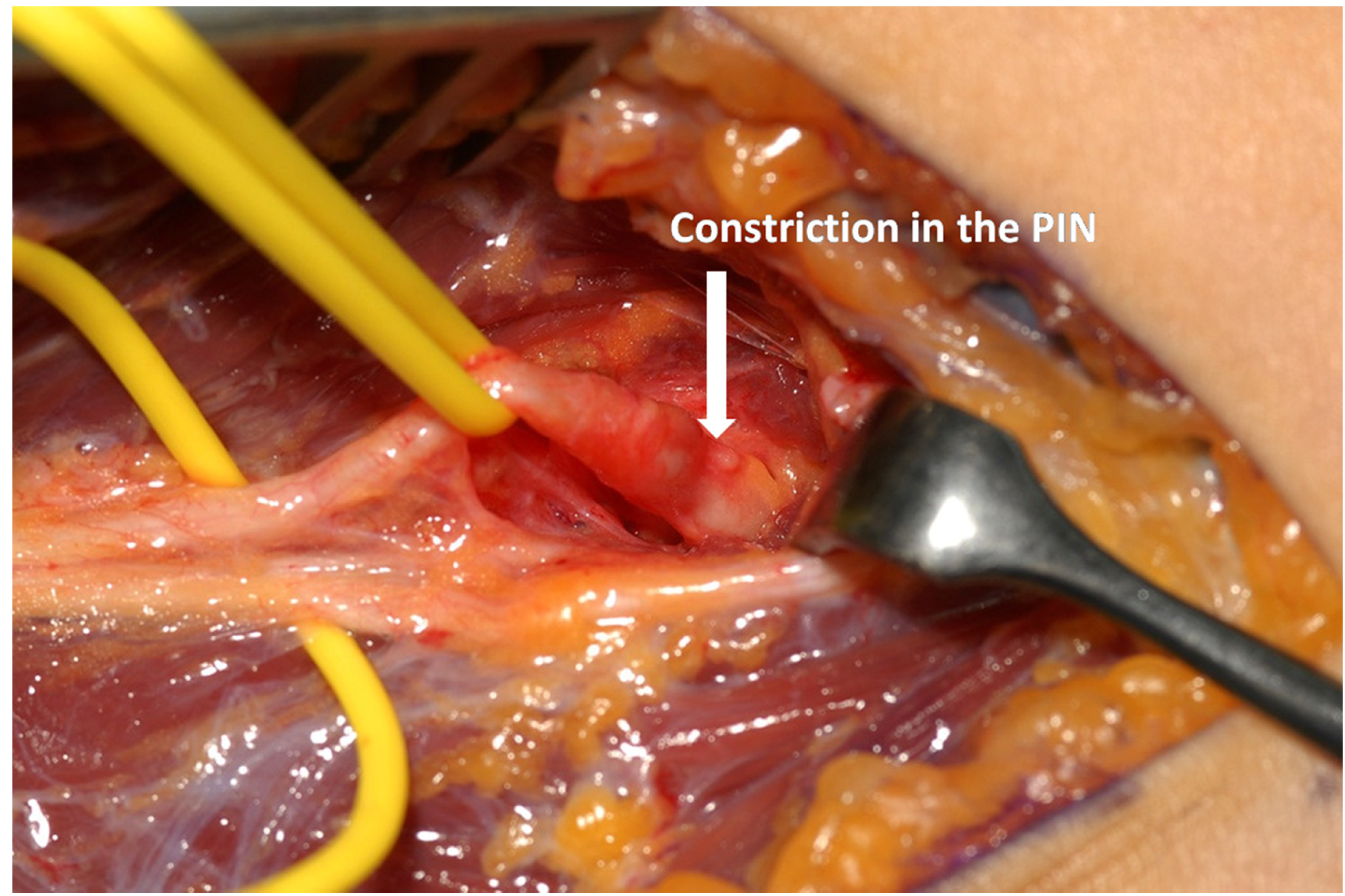
This approach is also favoured in PIN syndrome as the PIN is easy to find through this approach, before it passes under the arcade of Frohse, and it can be dissected with relative ease through to the distal edge of the supinator. Proximal dissection opens up the radial tunnel allowing for division of fascial bands and radio-recurrent vessels, or excision of any space occupying lesion which may be present. In fact it is seldom stated explicitly, but the surgery is mainly about dividing the superficial head of supinator over the PIN whilst providing an opportunity to check the other aetiologies that may compress the nerve.
Muscle-splitting approach
A longitudinal incision is made from the radial head along the muscle belly of brachioradialis. The fascia over brachioradialis is divided and the muscle fibres divided longitudinally to gain access to the radial tunnel, PIN and supinator muscle underneath the brachioradialis muscle.
Lateral approach
A 10 cm incision is made along the posterior border of brachioradialis muscle. The interval between brachioradialis muscle and ECRL is then opened, taking care for the posterior cutaneous nerve of the forearm which is found superficially between these two muscles. This gives good exposure to the PIN and SRN. Digital examination of the proximal radial nerve is performed to exclude a more proximal compression, which if present should then be released through a deep dissection [50].
Posterior (Henry or Thompson) approach
A 10 cm incision is made from the lateral epicondyle towards Lister’s tubercle. The posterior cutaneous nerve should be identified and protected. The interval between EDC and ECRB/ECRL is opened providing good exposure to the full length of the supinator muscle beneath. Suematsu and Hirayama [34] recommend this approach for type 2 (drop fingers) and type 3 (drop thumb) as compression of the medial and lateral branches of PIN can be easily assessed within or at the distal edge of the supinator. This approach is not suitable for accessing the radial tunnel.
Superficial radial nerve (SRN) compression
Cheiralgia paresthetica is the term Wartenberg coined for compression of the radial sensory nerve of the forearm after describing it in 5 cases in 1932. However it is now more commonly referred to as Wartenberg’s syndrome [51]. Patients complain of paraesthesia, pain and numbness over the dorso-radial aspect of the wrist in the distribution of the SRN. The SRN branches from the radial nerve proper within the radial tunnel at the level of the lateral epicondyle. It progresses distally deep to brachioradialis (BR) muscle and pierces through the deep fascia between the BR and ECRL tendons, around 8 cm proximal to the radial syloid, to enter the subcutaneous tissues. The nerve can be compressed during pronation when the tendons of the BR and ECRL approximate. Activities involving prolonged pronation, such as with writing or using a typewriter, or repetitive pronosupination, such as using a screwdriver, have been implicated in Wartenberg syndrome [52]. In 3–10% of individuals the SRN emerges through the tendon of BR [53], where it can also become entrapped [54]. Five centimeters proximal to the radial styloid the SRN bifurcates into a dorsal branch to the first webspace, and a more substantial radial branch to the dorsum of the thumb, which both run across the dorso-radial wrist. The nerve is susceptible to external compression against the radius in this superficial location, as has been reported with the wearing of wrist watches [55], and handcuffs [56]. In addition radial nerve branches may become entrapped in scar and irritated in distal radius fractures and the post-injury wrist deformity may further stretch the nerve during ulnar flexion. Other causes include diabetes, exposure to cold, iatrogenic, and proximal SRN neuropathies have been reported from lipomas and radial head fractures [57]. Over time oedema and fibrosis of the nerve result in compression, and ultimately Wallerian degeneration of the nerve [58].
Lanzetta and Foucher found that 50% of patients with Warternberg’s syndrome had associated De Quervain’s disease, but the two can be distinguished through appropriate clinical examination although overlap of symptoms exists [59]. Paraesthesia and a Tinel’s sign over the course of the nerve are positive in Wartenberg’s syndrome, and symptoms may be reproduced by pronation and ulnar wrist flexion, whereas Finklestein’s test is positive in De Quervain’s disease. Electrodiagnositic studies can be used to distinguish Wartenberg syndrome from De Quervain’s disease if required [58].
Treatment
In cases of external compression of the SRN, removing the cause should resolve the problem. When excessive pronosupination is implicated, avoidance or modification of aggravating activities and a local corticosteroid injection at the site of entrapment between the BR and ECRL tendons is generally successful. Splinting and rest will help, but is not usually tolerated because of the difficulty of splinting in forearm supination. Surgical release is rarely necessary. Lanzetta and Foucher report that conservative treatment was successful in 71% of cases, however they also found that 74% of those who did undergo surgery had excellent or good results [59].
Surgery
Prior to surgery the area where the Tinel’s sign is maximal is marked. A 2–3cm longitudinal incision is made over this area along the radial border of brachioradialis muscle. The nerve is identified and traced proximally, and the fascia between the tendons of BR and ECRL is opened to release the nerve as it passes through the deep fascia. A portion of the tendon of the BR may need to be resected to completely decompress the radial sensory nerve. Early movement of the forearm is encouraged postoperatively, and further desensitization of the radial sensory distribution on the dorsum of the hand may be required. Patients should avoid heavy lifting for the first month. Good results have also been reported with resection of a portion of the SRN such that the proximal stump lies beneath the BR muscle [55]. However, this results in permanent numbness to the radial sensory distribution of the hand, and risks painful neuroma formation, and is therefore not recommended.
References
[1] Latinovic R, Gulliford MC, Hughes RA. Incidence of common compressive neuropathies in primary care. J Neurol Neurosurg Psychiatry. 2006 Feb;77(2):263-5. DOI: 10.1136/jnnp.2005.066696[2] Abrams RA, Ziets RJ, Lieber RL, Botte MJ. Anatomy of the radial nerve motor branches in the forearm. J Hand Surg Am. 1997 Mar;22(2):232-7. DOI: 10.1016/S0363-5023(97)80157-8
[3] Thomas SJ, Yakin DE, Parry BR, Lubahn JD. The anatomical relationship between the posterior interosseous nerve and the supinator muscle. J Hand Surg Am. 2000 Sep;25(5):936-41. DOI: 10.1053/jhsu.2000.16360
[4] Lubahn JD, Cermak MB. Uncommon nerve compression syndromes of the upper extremity. J Am Acad Orthop Surg. 1998 Nov-Dec;6(6):378-86. DOI: 10.5435/00124635-199811000-00006
[5] Lister GD, Belsole RB, Kleinert HE. The radial tunnel syndrome. J Hand Surg Am. 1979 Jan;4(1):52-9. DOI: 10.1016/S0363-5023(79)80105-7
[6] Michele AA, Krueger FJ. Lateral epicondylitis of the elbow treated by fasciotomy. Surgery. 1956 Feb;39(2):277-84.
[7] Roles NC, Maudsley RH. Radial tunnel syndrome: resistant tennis elbow as a nerve entrapment. J Bone Joint Surg Br. 1972 Aug;54(3):499-508. DOI: 10.1302/0301-620X.54B3.499
[8] Spinner M. The arcade of Frohse and its relationship to posterior interosseous nerve paralysis. J Bone Joint Surg Br. 1968 Nov;50(4):809-12. DOI: 10.1302/0301-620X.50B4.809
[9] Henry AK, editor. Extensile exposure. Edinburgh: E and S. Livingstone Ltd.; 1957.
[10] Frohse F, Fränkel M. Die Muskeln des menschlichen Armes. Jena: G. Fisher; 1908.
[11] Hentz V. Invited discussion: proximal radial compression neuropathy. Annal Plast Surg. 2004;52(2):181-3. DOI: 10.1097/01.sap.0000095661.75866.38
[12] Arle JE, Zager EL. Surgical treatment of common entrapment neuropathies in the upper limbs. Muscle Nerve. 2000 Aug;23(8):1160-74.
[13] Barnum M, Mastey RD, Weiss AP, Akelman E. Radial tunnel syndrome. Hand Clin. 1996 Nov;12(4):679-89.
[14] Werner CO. Lateral elbow pain and posterior interosseous nerve entrapment. Acta Orthop Scand Suppl. 1979;174:1-62. DOI: 10.3109/ort.1979.50.suppl-174.01
[15] Prasartritha T, Liupolvanish P, Rojanakit A. A study of the posterior interosseous nerve (PIN) and the radial tunnel in 30 Thai cadavers. J Hand Surg Am. 1993 Jan;18(1):107-12. DOI: 10.1016/0363-5023(93)90253-Y
[16] Werner CO, Haeffner F, Rosén I. Direct recording of local pressure in the radial tunnel during passive stretch and active contraction of the supinator muscle. Arch Orthop Trauma Surg. 1980;96(4):299-301. DOI: 10.1007/BF00432870
[17] Sarhadi NS, Korday SN, Bainbridge LC. Radial tunnel syndrome: diagnosis and management. J Hand Surg Br. 1998 Oct;23(5):617-9.DOI: 10.1016/S0266-7681(98)80015-6
[18] Loh YC, Lam WL, Stanley JK, Soames RW. A new clinical test for radial tunnel syndrome--the Rule-of-Nine test: a cadaveric study. J Orthop Surg (Hong Kong). 2004 Jun;12(1):83-6.DOI: 10.1177/230949900401200115
[19] Stanley J. Radial tunnel syndrome: a surgeon's perspective. J Hand Ther. 2006 Apr-Jun;19(2):180-4. DOI: 10.1197/j.jht.2006.02.005
[20] Dang AC, Rodner CM. Unusual compression neuropathies of the forearm, part I: radial nerve. J Hand Surg Am. 2009 Dec;34(10):1906-14. DOI: 10.1016/j.jhsa.2009.10.016
[21] Kupfer DM, Bronson J, Lee GW, Beck J, Gillet J. Differential latency testing: a more sensitive test for radial tunnel syndrome. J Hand Surg Am. 1998 Sep;23(5):859-64. DOI: 10.1016/S0363-5023(98)80163-9
[22] Rosén I, Werner CO. Neurophysiological investigation of posterior interosseous nerve entrapment causing lateral elbow pain. Electroencephalogr Clin Neurophysiol. 1980 Oct;50(1-2):125-33. DOI: 10.1016/0013-4694(80)90329-6
[23] Rosenberg ZS, Bencardino J, Beltran J. MR features of nerve disorders at the elbow. Magn Reson Imaging Clin N Am. 1997 Aug;5(3):545-65.
[24] Grant GA, Britz GW, Goodkin R, Jarvik JG, Maravilla K, Kliot M. The utility of magnetic resonance imaging in evaluating peripheral nerve disorders. Muscle Nerve. 2002 Mar;25(3):314-31. DOI: 10.1002/mus.10013
[25] Atroshi I, Johnsson R, Ornstein E. Radial tunnel release. Unpredictable outcome in 37 consecutive cases with a 1-5 year follow-up. Acta Orthop Scand. 1995 Jun;66(3):255-7. DOI: 10.3109/17453679508995536
[26] Lawrence T, Mobbs P, Fortems Y, Stanley JK. Radial tunnel syndrome. A retrospective review of 30 decompressions of the radial nerve. J Hand Surg Br. 1995 Aug;20(4):454-9. DOI: 10.1016/S0266-7681(05)80152-4
[27] Moss SH, Switzer HE. Radial tunnel syndrome: a spectrum of clinical presentations. J Hand Surg Am. 1983 Jul;8(4):414-20. DOI: 10.1016/S0363-5023(83)80201-9
[28] Ritts GD, Wood MB, Linscheid RL. Radial tunnel syndrome. A ten-year surgical experience. Clin Orthop Relat Res. 1987 Jun;(219):201-5.
[29] Guillain G, Courtellemont R. Role of the supinator in radial nerve paralysis: pathogenesis of a partial radial nerve paralysis in an orchestra conductor. Presse Medicale. 1905;7:50–52.
[30] Kruse F Jr. Paralysis of the dorsal interosseous nerve not due to direct trauma; a case showing spontaneous recovery. Neurology. 1958 Apr;8(4):307-8. DOI: 10.1212/WNL.8.4.307
[31] Silverstein A. Progressive paralysis of the dorsal interosseous nerve. Arch Neurol Psychiatry. 1937;38:885.
[32] Hobhouse NH. A case of posterior interosseous paralysis. BMJ. 1936;1:841.
[33] Green DP, Hotchkiss RN, Pederson WC, Wolfe SW, editors. Green's operative hand surgery. 3rd ed. New York: Churchill Livingstone; 1993.
[34] Suematsu N, Hirayama T. Posterior interosseous nerve palsy. J Hand Surg Br. 1998 Feb;23(1):104-6. DOI: 10.1016/S0266-7681(98)80234-9
[35] Blakemore ME. Posterior interosseous nerve paralysis caused by a lipoma. J R Coll Surg Edinb. 1979 Mar;24(2):113-6.
[36] Ganapathy K, Winston T, Seshadri V. Posterior interosseous nerve palsy due to intermuscular lipoma. Surg Neurol. 2006 May;65(5):495-6; discussion 496. DOI: 10.1016/j.surneu.2005.06.035
[37] Fitzgerald A, Anderson W, Hooper G. Posterior interosseous nerve palsy due to parosteal lipoma. J Hand Surg Br. 2002 Dec;27(6):535-7. DOI: 10.1054/jhsb.2002.0783
[38] Wu KT, Jordan FR, Eckert C. Lipoma, a cause of paralysis of deep radial (posterior interosseous) nerve: report of a case and review of the literature. Surgery. 1974 May;75(5):790-5.
[39] Mass DP, Tortosa R, Newmeyer WL, Kilgore ES Jr. Compression of posterior interosseous nerve by a ganglion--case report. J Hand Surg Am. 1982 Jan;7(1):92-4. DOI: 10.1016/S0363-5023(82)80022-1
[40] Tonkin MA. Posterior interosseous nerve axonotmesis from compression by a ganglion. J Hand Surg Br. 1990 Nov;15(4):491-3. DOI: 10.1016/0266-7681(90)90100-I
[41] Bowen TL, Stone KH. Posterior interosseous nerve paralysis caused by a ganglion at the elbow. J Bone Joint Surg Br. 1966 Nov;48(4):774-6. DOI: 10.1302/0301-620X.48B4.774
[42] Steiger R, Vögelin E. Compression of the radial nerve caused by an occult ganglion. Three case reports. J Hand Surg Br. 1998 Jun;23(3):420-1. DOI: 10.1016/S0266-7681(98)80073-9
[43] Hashizume H, Inoue H, Nagashima K, Hamaya K. Posterior interosseous nerve paralysis related to focal radial nerve constriction secondary to vasculitis. J Hand Surg Br. 1993 Dec;18(6):757-60. DOI: 10.1016/0266-7681(93)90239-C
[44] Marmor L, Lawrence JF, Dubois EL. Posterior interosseous nerve palsy due to rheumatoid arthritis. J Bone Joint Surg Am. 1967 Mar;49(2):381-3. DOI: 10.2106/00004623-196749020-00020
[45] Kato H, Iwasaki N, Minami A, Kamishima T. Acute posterior interosseous nerve palsy caused by septic arthritis of the elbow: a case report. J Hand Surg Am. 2003 Jan;28(1):44-7. DOI: 10.1053/jhsu.2003.50017
[46] Gutowski KA. Hand II: peripheral nerves and tendon transfers. Selected readings in Plastic surgery. Dallas: University of Texas Southwestern Medical Center and Baylor University Medical Center. 2003;9(33):1-39.
[47] Jebson PJ, Engber WD. Radial tunnel syndrome: long-term results of surgical decompression. J Hand Surg Am. 1997 Sep;22(5):889-96. DOI: 10.1016/S0363-5023(97)80086-X
[48] De Smet L, Van Raebroeckx T, Van Ransbeeck H. Radial tunnel release and tennis elbow: disappointing results? Acta Orthop Belg. 1999 Dec;65(4):510-3.
[49] Vrieling CR, P. Geertzeen, J. Posterior interosseous nerve sydrome: literature review and report of 14 cases. Eur J Plast Surg. 1998;21:196-202. DOI: 10.1007/s002380050068
[50] Wolfe SW, Pederson WC, Hotchkiss RN, Kozin SH, editors. Green's operative hand surgery. 6th ed. New York: Churchill Livingston; 2010.
[51] Ehrlich W, Dellon AL, Mackinnon SE. Classical article: Cheiralgia paresthetica (entrapment of the radial nerve). A translation in condensed form of Robert Wartenberg's original article published in 1932. J Hand Surg Am. 1986 Mar;11(2):196-9. DOI: 10.1016/S0363-5023(86)80050-8
[52] Dellon AL, Mackinnon SE. Radial sensory nerve entrapment in the forearm. J Hand Surg Am. 1986 Mar;11(2):199-205. DOI: 10.1016/S0363-5023(86)80051-X
[53] Abrams RA, Brown RA, Botte MJ. The superficial branch of the radial nerve: an anatomic study with surgical implications. J Hand Surg Am. 1992 Nov;17(6):1037-41. DOI: 10.1016/S0363-5023(09)91056-5
[54] Turkof E, Puig S, Choi SS, Zöch G, Dellon AL. The radial sensory nerve entrapped between the two slips of a split brachioradialis tendon: a rare aspect of Wartenberg's syndrome. J Hand Surg Am. 1995 Jul;20(4):676-8. DOI: 10.1016/S0363-5023(05)80290-4
[55] Braidwood AS. Superficial radial neuropathy. J Bone Joint Surg Br. 1975 Aug;57(3):380-3. DOI: 10.1302/0301-620X.57B3.380
[56] Grant AC, Cook AA. A prospective study of handcuff neuropathies. Muscle Nerve. 2000 Jun;23(6):933-8. DOI: 10.1002/(SICI)1097-4598(200006)23:6<933::AID-MUS14>3.0.CO;2-G
[57] Tosun N, Tuncay I, Akpinar F. Entrapment of the sensory branch of the radial nerve (Wartenberg's syndrome): an unusual cause. Tohoku J Exp Med. 2001 Mar;193(3):251-4. DOI: 10.1620/tjem.193.251
[58] Green DP, Wolfe SW, Pederson WC, Hotchkiss RN, Wolfe WS, editors. Green's Operative Hand Surgery. 5th ed. Philadelphia: Elsevier Churchill Livingstone; 2005.
[59] Lanzetta M, Foucher G. Entrapment of the superficial branch of the radial nerve (Wartenberg's syndrome). A report of 52 cases. Int Orthop. 1993 Dec;17(6):342-5. DOI: 10.1007/BF00180450



