Ulnar nerve compression
Raymond Anakwe 1
1 Upper Limb Surgery, Wrightington Hospital, Wigan, United Kingdom
Abstract
Although compression of the ulnar nerve is a common condition, many controversies remain in its management. Compression of the ulnar nerve around the elbow, cubital tunnel syndrome, is most common. This chapter reviews the relevant anatomy and the pathophysiology of ulnar nerve entrapment. Diagnosis depends on clinical examination and neurophysiology tests. Non-operative management includes activity modification and splintage. Surgery is recommended for patients with persisting symptoms. In situ decompression of the ulnar is adequate in most cases, with anterior transposition being reserved for recurrent symptoms. The ulnar nerve may also be compressed at the wrist requiring surgical decompression.
Introduction
Entrapment of the ulnar nerve is a common condition that raises many controversial questions. The most common site of compression of the ulnar nerve is around the elbow, especially at the leading edge of flexor carpi ulnaris, although other potential sites of compression have been identified. An understanding of the anatomy of the nerve can aid diagnosis and consideration of the pathophysiology of ulnar nerve entrapment can guide management.
Basics
Anatomy of ulnar nerve
The ulnar nerve is a mixed motor and sensory nerve arising from the ventral rami of C8 and T1 roots of the brachial plexus. It is a terminal branch of the medial cord although it may also receive additional nerve fibres from the lateral cord of the brachial plexus, often from the anterior ramus of the C7 nerve root [1]. In the upper arm the nerve lies postero-medial to the brachial artery. Classically the nerve is described as passing posteriorly through the medial intermuscular septum in the midportion of the arm, however in one anatomical study the nerve lay in the posterior compartment throughout its course in 8/10 cadavers examined [2]. When travelling on the posterior surface of the medial intermuscular septum it is said to be covered by a thickened band of fibrous tissue extending from the medial head of triceps to the medial intermuscular septum, the Arcade of Struthers. The existence of the arcade of Struthers is questioned by some authors [2]. In cadaveric studies that report the presence of a fibrous band over the ulnar nerve the prevalence varies, with quoted rates of 13.5% [3], 68% [4] and 100% [5], raising concerns about observer bias.
The nerve passes posterior to the medial epicondyle of the humerus in the fibro-osseous cubital tunnel, the floor made up by the humerus, joint capsule, posterior and transverse band of the medial collateral ligament superiorly and laterally, and the roof by the cubital retinaculum. This structure, which is tight in elbow flexion and lax in extension, has varied anatomy. O’Driscoll examined 27 frozen cadavers, in one the retinaculum was absent and the nerve subluxed (type 0), in 17 the nerve was covered by a thin retinaculum that O’Driscoll reported as “normal” (type 1a), six of 27 had a “pathologically thickened” retinaculum with reported signs of nerve compression (type 1b), and in three the retinaculum was replaced by muscle fibres of anconeus epitrochlearis (type 2) [6]. The retinaculum is thought to be a remnant of the anconeus epitrochlearis and serves to maintain the ulnar nerve in its bed during elbow flexion.
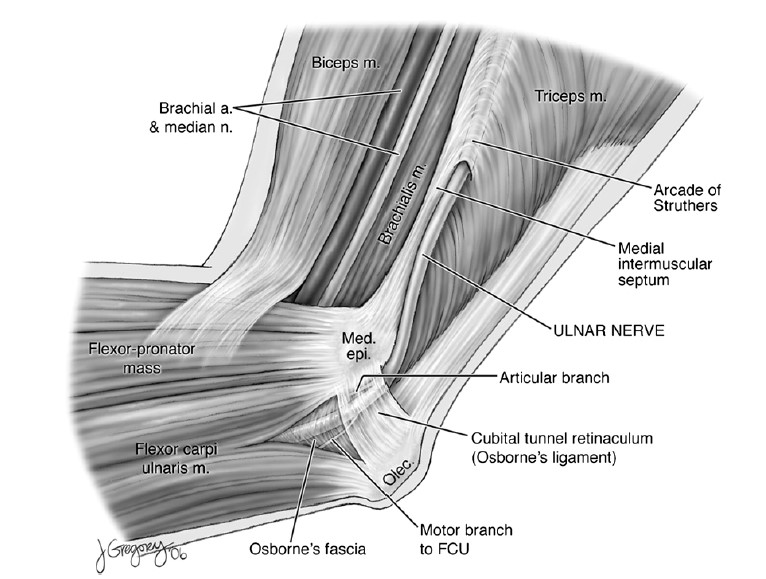
Immediately after exiting from beneath the cubital retinaculum the nerve passes under the deep aponeurosis of the flexor carpi ulnaris (FCU). This is identified between the ulnar and humeral heads of FCU and has been shown to extend up to 9 cm distal to the retrocondylar groove [7]. In the same study the aponeurosis has been shown to have either three or four fascial thickenings that the authors felt were possible sites for nerve entrapment. Incisions around the medial epicondyle for decompression or transposition of the ulnar nerve will cross branches of the medial antebrachial cutaneous nerve, a direct branch of the medial cord (C8, T1) [1]. In over half of examined specimens the posterior branch has been shown to pass over the ulnar nerve at or within 2 cm proximal to the medial epicondyle [8].
The ulnar nerve enters the forearm between the ulnar and humeral heads of the flexor carpi ulnaris (FCU) muscle, travelling in the flexor compartment of the forearm beneath the FCU and running on the flexor digitorum profundus (FDP) muscle, medial to the ulnar artery. As the ulnar nerve courses distally in the forearm, there is a rotational reorientation of the nerve fibres so that the motor fibres initially lying dorsal and radial come to lie more volar and ulnar. Approximately 5cm proximal to the wrist crease it gives off a dorsal cutaneous branch which runs medially and dorsally between the FCU tendon and the distal ulna to supply sensation to the dorsum of the ulnar border of the hand and the dorsal surfaces of the ulnar one and a half digits. The main ulnar nerve passes medially at the wrist joint across the flexor retinaculum medial to the FCU tendon and over the pisohamate ligament between the palpable pisiform and the hamate bones and deep to the volar carpal ligament. This osseo-ligamentous area is defined as distal ulnar tunnel and here the ulnar nerve divides again into a superficial branch running on or in the muscles of the hypothenar eminence and a deep branch curving around the hook of the hamate. The superficial branch supplies motor innervations to the palmaris brevis (PB) muscle before terminating as the digital nerves to the little and ring fingers. The deep branch of the ulnar nerve curves around the hook of the hamate passing deep to the muscles of the hypothenar eminence and supplying motor innervation to the hypothenar eminence, adductor pollicis (AD) muscle, the interossei, the medial two lumbricals and the deep head of flexor pollicis brevis (FPB).
Branches of the ulnar nerve
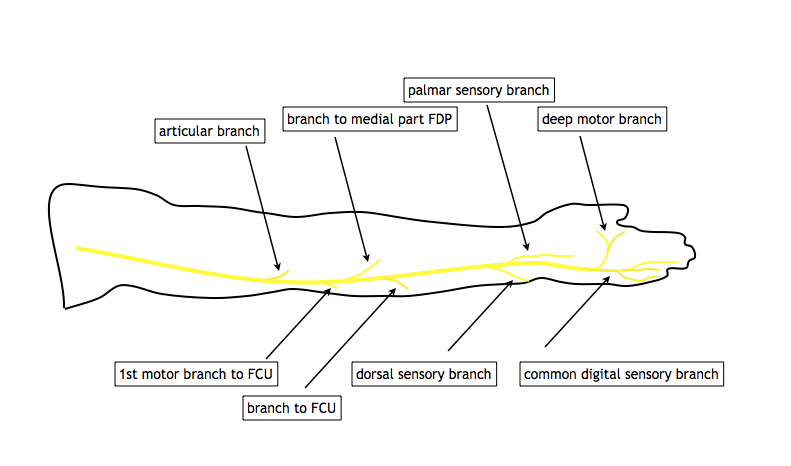
The ulnar nerve does not typically give off any branches in the arm. At the level of the elbow, where it runs behind the medial epicondyle, the nerve gives off at least one and occasionally more articular branches to the elbow and just distal to this, the first main branch of the nerve supplies the FCU muscle. As it runs on the FDP muscle, the ulnar nerve supplies motor innervation to the ulnar half of this muscle and just proximal to the wrist joint it gives of a small superficial volar branch which emerges medial to FCU, penetrates the deep fascia and supplies sensation to the ulnar palm. The terminal branches of the ulnar nerve are the dorsal cutaneous branch proximal to the wrist joint and the more distal superficial and deep divisions of the ulnar nerve.
While this represents the typical anatomy of the ulnar nerve, a number of variations have been described. In particular, the median nerves and ulnar nerves in the forearm and palm may share anastomoses so that the usual motor innervations of one nerve may be supplied by the other. This and other variations to the classically described anatomy of these nerves may cause diagnostic delay and confusion in some cases of compressive neuropathy or nerve injury.
Potential sites of compression
The ulnar nerve may be susceptible to compression at a number of sites along its course. Compression at or around the elbow, particularly in the cubital tunnel, is more common than compression at the level of the wrist.
Cubital tunnel syndrome
Cubital tunnel syndrome is the second most common entrapment neuropathy in the upper limb after carpal tunnel syndrome. The syndrome consists of a collection of symptoms and signs in the ulnar nerve distribution of the hand; that is cutaneous innervation of the little and ulnar half of the ring finger, and motor innervation to the small muscles of the hand, flexor carpi ulnaris and flexor digitorum profundus to the ring and little fingers. Altered sensation on the dorsum of the hand in the area supplied by the dorsal cutaneous branch of the ulnar nerve can help differentiate a “high” lesion of the nerve from a “low” lesion in Guyon’s canal. The “ulna paradox” describes the different motor loss patterns, in which a low lesion will typically present with more marked clawing of the little and ring fingers due to the unopposed action of the long flexors, whereas in a high lesion around the elbow the clawing is more mild as the long flexors are affected too.
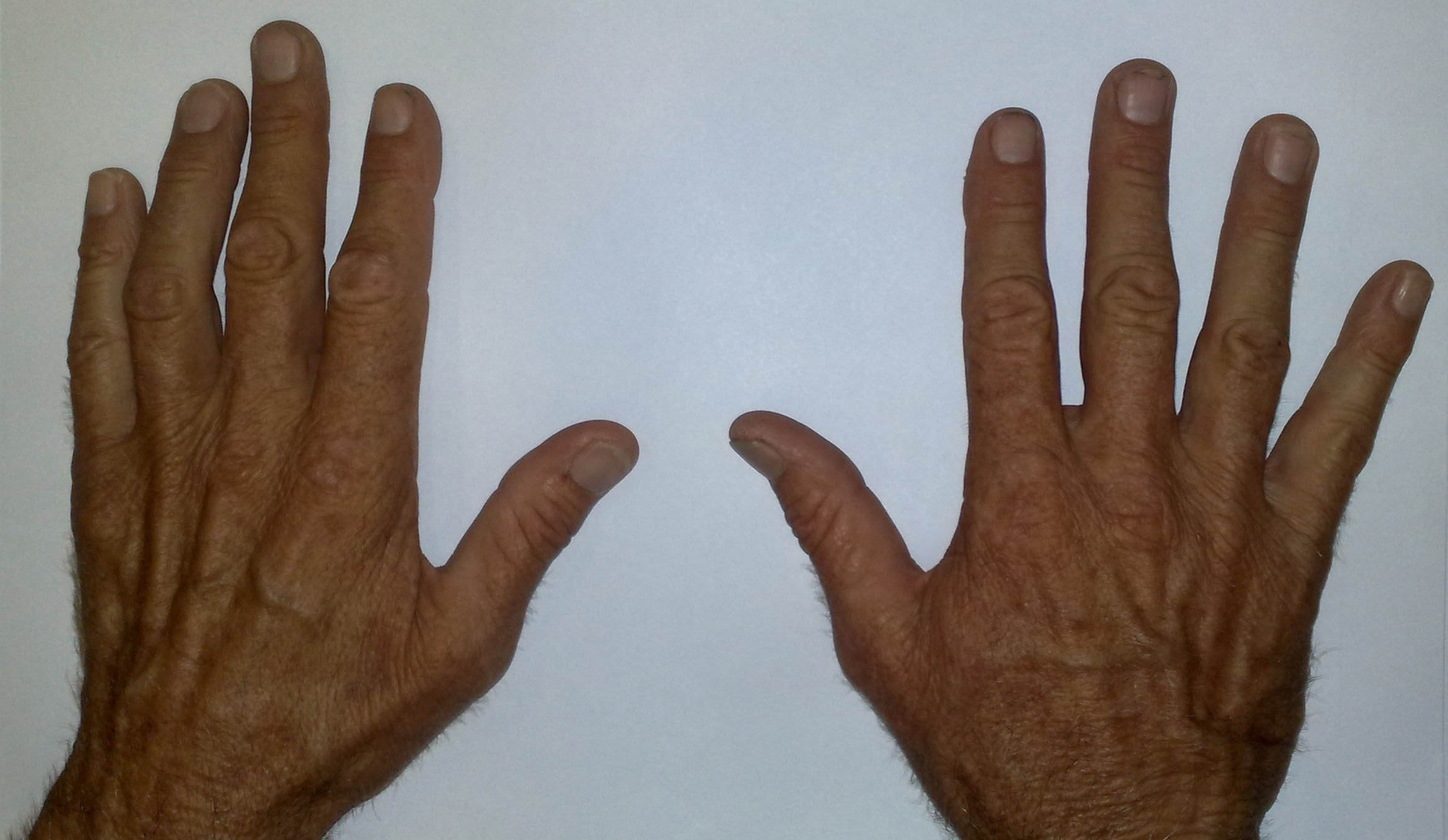
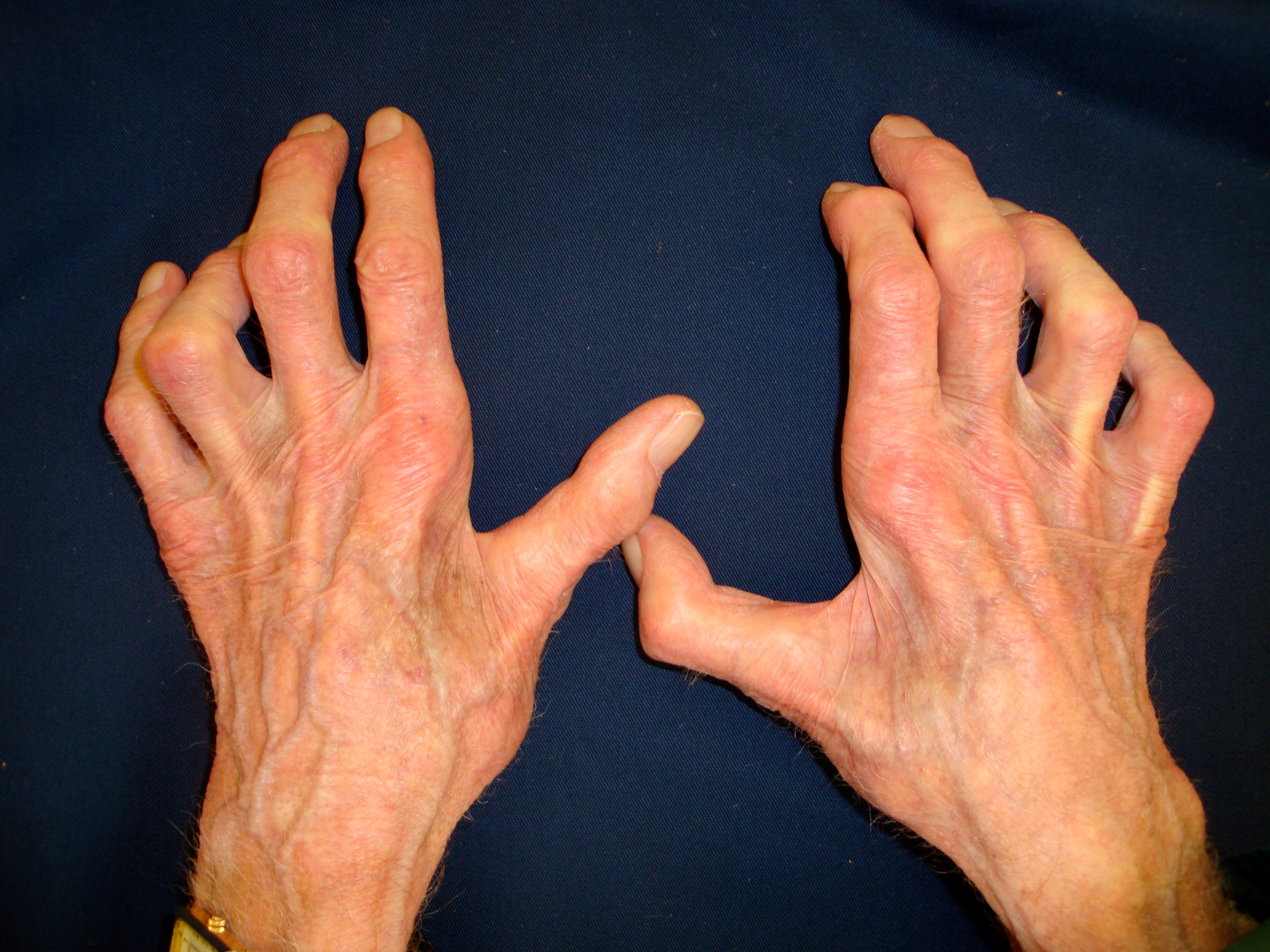
Aetiology of cubital tunnel syndrome
Historically surgeons have debated whether the syndrome is caused by compression of the ulnar nerve or by traction. A number of studies have been performed in cadavers to examine the hypothesis that the ulnar nerve is compressed by overlying structures, mainly the cubital retinaculum, during elbow flexion. Vanderpool [9] demonstrated that the distance between insertion and origin of the cubital retinaculum increases on elbow flexion, and it has been assumed that this must be associated with a decrease in the space available for the nerve and compression. Most studies show significant increases in extra-neural pressure from around 6mmHg to as high as 65mmHg in full flexion [10], [11], and intraneural pressures have been shown to rise too [12]. It is not clear whether this is due to compressive forces, nor whether this causes ulnar neuropathy. In clinical studies similar extra-neural pressure changes are seen, and have been shown to correlated with stage of disease and mean conduction velocity on neurophysiological examination [13]. The authors were unable to conclude that compression was the major factor in the aetiology of ulnar neuritis. More recent work by the same group has shown that extra-neural pressures are higher in osteoarthritic elbows, which the authors argue supports role of compression in cubital tunnel syndrome [14].
Strain (change in length of the nerve) in the ulnar nerve with elbow flexion has been suggested as a possible aetiology. Proponents argue that traction is applied to the nerve with increasing elbow flexion as the nerve passes posterior to the axis of flexion of the elbow, and therefore surgical treatment must aim to reduce the traction by anterior transposition or epicondylectomy. Laboratory studies have demonstrated that the strain in the nerve increases with maximal elbow flexion and that the highest strain is located posterior to the medial epicondyle [15]. The authors argue that the strains recorded are equivalent to the strains noted to cause alteration in nerve conduction in animal studies [16]. Other studies indicate that while there is some elongation significant change in length of the nerve do not occur until flexion exceeds 90° [17]. Neither of these studies take account of stress relaxation that occurs in connective tissue and has been shown to be associated with a return to normal conduction patterns [18]. Further evidence to support traction rather than compression as the predominant force across the ulnar nerve at the elbow comes from the work of Gelberman [19]. In this study magnetic resonance images of the elbow were obtained in varying degrees of flexion. Pressure transducers were also inserted transdermally under ultrasound guidance to measure intra- and extra-neural pressures. This study reported that while the shape and area of the cubital tunnel did decrease with flexion, the cross sectional area of the ulnar nerve decreased proportionally more. Thus the proportional area available for the nerve actually increased. In addition the intraneural pressure increased faster and was higher than the extraneural pressure with flexion and was not significantly altered by simple nerve decompression. The authors concluded that traction is a more significant force on the ulnar nerve at the elbow than compression.
One further factor that must be considered is friction. Ulnar nerve subluxation is not uncommon in the normal population [20] and has been reported in 7% of patients undergoing surgery for cubital tunnel syndrome [21]. It has been proposed that repeated subluxation can result in a friction neuritis of the nerve, or results in the nerve being more exposed to repetitive trauma lying over the bony prominence of the medial epicondyle. It is not clear yet which of these factors is responsible and as with many syndromes the aetiology may be multifactorial.
Classification of cubital tunnel syndrome
McGowan proposed a grading system for cubital tunnel syndrome into three categories based purely on motor function that was later modified to include sensory findings; grade 1: sensory symptoms only, grade 2: sensory alteration with muscle weakness, grade 3: muscle paresis [22]. Dellon has also described a three level grading system that is widely used (Table 1) [23].
| Stage | Sensory | Motor | Tests |
|---|---|---|---|
| Mild | Intermittent paraesthesias, vibratory sensation increased | Subjective weakness, clumsiness or loss of coordination | Elbow flexion test or Tinel's sign positive |
| Moderate | Persistent paraesthesias, vibratory perception normal or decreased | Measurable weakness in pinch and grip | Elbow flexion test or Tinel's sign positive, finger crossing may be abnormal |
| Severe | Persistent paraesthesias, vibratory perception decreased, abnormal two point discrimination (static >6mm, moving >4mm) | Measurable weakness in pinch and grip plus muscle atrophy | Elbow flexion test or Tinel's sign positive, finger crossing abnormal |
Clinical aspects
The reported age standardised incidence rate of cubital tunnel syndrome in UK general practice is 170.4 per 100,000 females and is 38% more common in males at a rate of 235.0 per 100,000 [24]. The peak incidence in males is in those aged 55 to 64, in females the incidence peaks a decade earlier (those aged 45 to 54). Only thirty percent of affected individuals were reported to undergo operative treatment.
Although work related activities involving repetitive elbow flexion and extension might aggravate cubital tunnel syndrome, no scientific data have supported work as a causal factor. Musicians and sportsmen have been reported to be at risk [25].
Examination
Findings include sensory involvement on the ulnar one and half digits and the ulnar dorsal aspect of the hand suggests cubital tunnel syndrome, as the dorsal cutaneous branch of the ulnar nerve originates proximal to the canal of Guyon. Only rarely do patients present with wasting of the intrinsic musculature in the hand. The Semmes Weinstein monofilament test and vibration testing are helpful in detecting sensory impairment during the earlier stages of the disease. For more severe cases, static and moving 2–point discrimination can be useful. Comparison with the asymptomatic arm can identify weakness of the intrinsic musculature. Patients may also complain of pain at the elbow and hypersensitivity with palpation of the ulnar nerve as it travels around the elbow.
Special tests
Several tests exist to clinically diagnose cubital tunnel syndrome.
Tinel’s test: This is supposed to indicate axonal damage. At the elbow, it consists of gentle tapping 4–6 times just proximal to the cubital tunnel. The presence or absence of a tingling sensation in the hand is noted. This test is not particularly sensitive or specific and is really only useful when considered together with other tests. Indeed nearly 24% of asymptomatic people have this finding [26].
Elbow flexion test: The elbow flexion test is performed as follows: The patient should sit with both arms and shoulders in the anatomic position and free from any external sources of compression. Both elbows should be fully flexed and both wrists fully extended. Radial pulses should be checked to ensure adequate perfusion. In theory this test maximises both compressive and tensile forces on the nerve. Patients are then asked at regular intervals for a total period of three minutes whether pain, numbness or tingling occurred or increased. The location of symptoms should be noted and the severity graded as 1 (minimal) to 3 (severe). This test as described by Buehler and Thayer demonstrated pain followed by numbness and then tingling in order of incidence [27].
The pressure provocation test: This test involves the examiner’s long and index fingers being placed on the patient’s ulnar nerve immediately proximal to the cubital tunnel with the elbow in 20° flexion and full forearm supination. Pressure is applied for 60 seconds bilaterally and the symptoms recorded at 30 and 60 seconds. It’s considered positive if the patient reports symptoms located within the distribution of the ulnar nerve.
The pressure and flexion provocative test: Both can be combined to increase the sensitivity, specificity and positive predictive value [28].
Diagnostics
The most useful investigations are neurophysiological studies including nerve conduction studies and electromyography. Motor nerve conduction studies are considered the most useful tests for localising nerve damage at the elbow [29]. The aim of electrodiagnosis is to confirm ulnar nerve dysfunction, localise the region of entrapment and determine the severity of nerve impairment [30], [31], [32] (see also Basics in clinical neurophysiology).
The most common localising sign is that of slowed motor conduction velocity (Kothari et al., 1998) [29]. Padua et al., 2001 found that this did reveal false negatives though, in those with milder symptoms [32]. Kothari et al., 1998 also found that sensitivity was increased when looking at the first dorsal interosseous rather than abductor digiti minimi [29]. Other authors postulate that recordings are similar, however there is a greater yield from the first dorsal interosseous [31]. In comparing the relative slowing of the conduction velocities of the first dorsal interosseous with that of the abductor digiti minimi, a specificity of 100% was achieved [33]. The abductor pollicis brevis should also be examined to exclude a C8-T1 nerve root or inferior brachial plexus lesion.
Plain radiographs are useful in the patient with arthritis, previous trauma or abnormal carrying angle. Chest radiographs should be considered to exclude a Pancoast tumour whenever a history of smoking, ulnar nerve symptoms and shoulder pain is reported.
Ultrasonography and MRI have been used as an aid in diagnosis [34]. Britz et al. showed a strong correlation with positive MRI finding, such as an increase in signal around the nerve and nerve compression [35]. Indeed they felt MRI was more sensitive in diagnosing ulnar compression than electrodiagnostic studies.
Therapy
Non-operative management
Knowledge of the natural history of a condition is important when considering treatment alternatives. Padua et al. reported that approximately half of patients with positive neurophysiology finding of cubital tunnel syndrome reported improvement in symptoms at 12 months without any specific intervention. The authors acknowledged however that most patients had adopted posture changes after diagnosis and this may have influenced the reported outcome [36]. Non-operative treatment has been proposed for patients with sensory symptoms only, with 89% of patients managed effectively without surgical intervention in one series [23]. The goals are to eliminate the frequency of symptoms and prevent further progression. Interventions include patient education, activity modification and splintage. Seror demonstrated 80–90% improvement both in subjective symptoms and neurophysiology with night splinting alone [37]. Svernlov et al. performed a randomised trial to compare night splinting, nerve gliding exercises and a control group who were given information about the condition but no active treatment. They reported no significant differences in outcome between the three groups and concluded that for patients with mild or moderate symptoms the prognosis is good with simple information and activity modification [38].
Operative management
Operative treatment may be indicated in those for whom symptoms have persisted for longer than six months, and those who have failed conservative therapy. Symptoms ongoing for longer than this tend to have a worse prognosis regardless of management. Dellon et al. reported that the need for surgical treatment was 21% within 6 years for those with mild symptoms, 33% within 3 years for moderate symptoms, and 62% within 3 years for severe symptoms [39].
The first description of surgical intervention for ulnar nerve compression at the elbow was from Henry Earle in London in 1816. He described the relief of severe pain in the ulnar nerve distribution in the hand of a 14 year old girl by sectioning of the ulnar nerve above the elbow. This resulted unsurprisingly in a dense nerve palsy. Subsequently treatment with anterior intramuscular transposition was described by Adson in 1918, then in 1942 Learmonth described the submuscular transposition. In-situ decompression was described by Osborne in 1957, who also gave his name to the cubital retinaculum and deep fascia of flexor carpi radialis. More recently endoscopic techniques for in-situ decompression have been reported. King and Morgan described medial epicondylectomy in 1958 as a way of reducing the tension in the nerve.
In-situ decompression
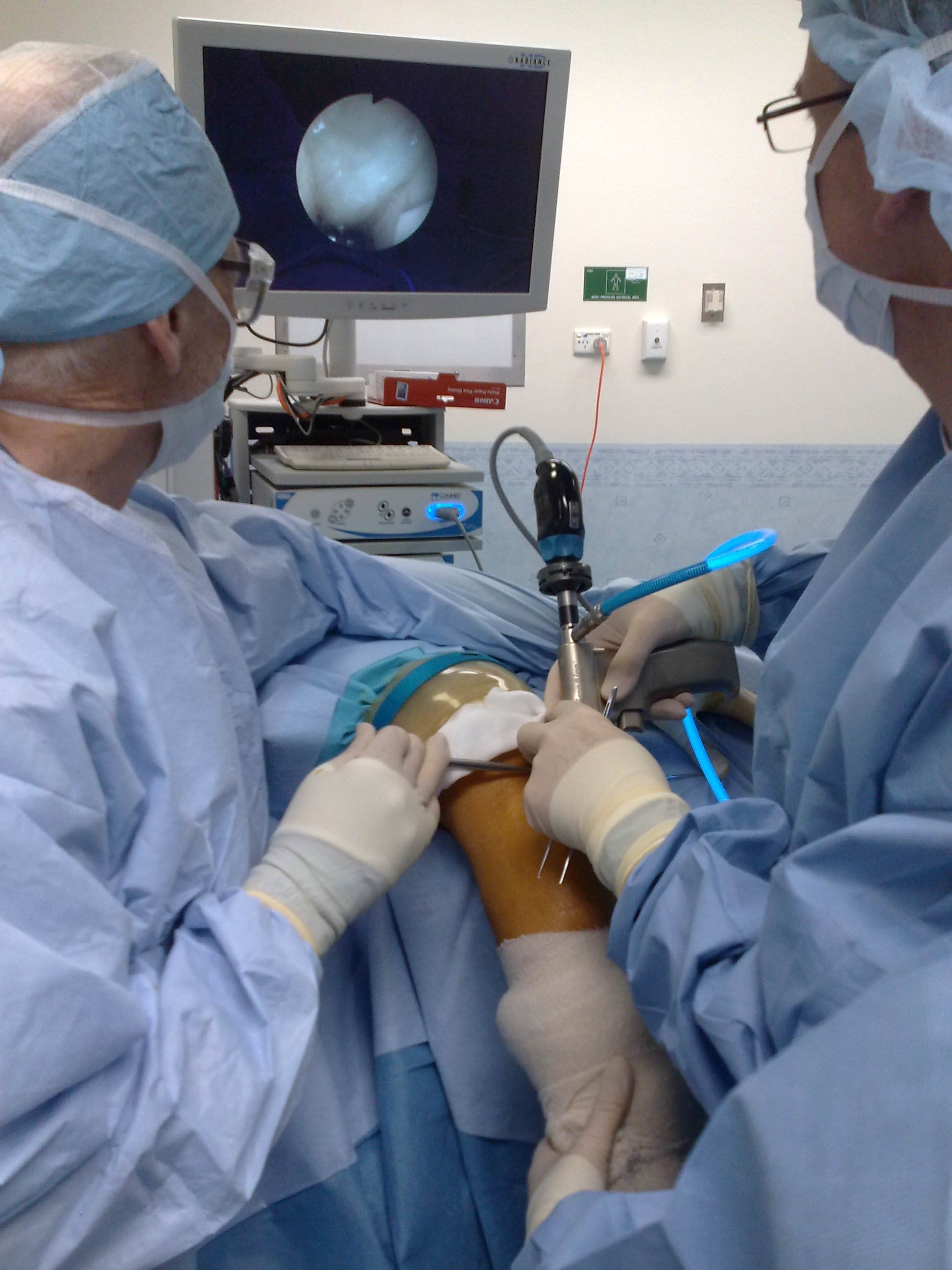
Cohort studies have reported satisfactory outcomes after in situ decompression [40]. In situ decompression can be performed through a standard incision (from 5 cm proximal to 5 cm distal to the medial epicondyle) or through a mini-open approach (less than 2.5 cm) [41], [42]. The latter facilitated by placing the patient in a lateral position with the elbow bent over a bolster. Care must be taken to avoid injury to the posterior branch of the medial cutaneous nerve of the forearm that will cross the wound approximately 2 cm proximal to the medial epicondyle. It is important to decompress all layers covering the nerve, particularly the deep fascia of flexor carpi ulnaris (Osborne’s Fascia) that is a common source of ulnar nerve entrapment. It is not necessary to dissect around the nerve once all covering layers have been divided. This will only increase the risk of nerve instability or disruption of the blood supply to the nerve. In patients with an anconeus epitrochlearis the muscle fibres should be excised, not simply divided, to prevent recurrence. Stability of the nerve can be assessed by flexion of the elbow on the operating table. Several techniques for endoscopic in-situ decompression have been described [43], [44], [45], [46]. All studies have found that endoscopic release is a safe and reliable treatment for the condition, particularly in patients with mild to moderate symptoms. A comparative study of open and endoscopic in-situ decompression reported equivalent outcomes between the two groups. The authors reported that the complication rate was higher in the open group when issues such a scar tenderness and medial elbow pain or numbness were included [47].
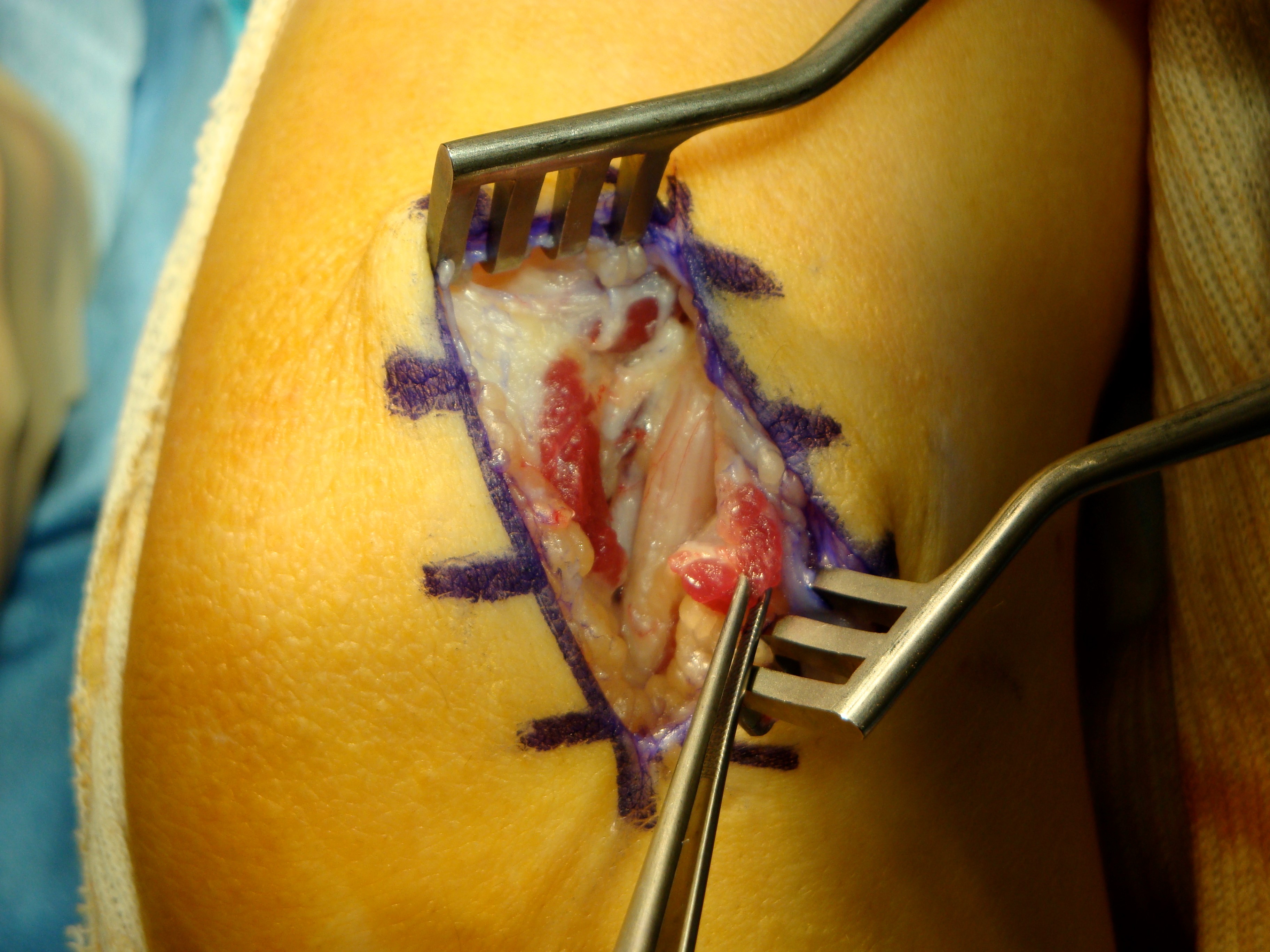
Anterior transposition
Anterior transposition of the ulnar nerve is generally reserved for patients with an unstable nerve or with recurrent symptoms after successful in-situ decompression. Several techniques for anterior transposition have been described, subcutaneous, subfascial and submuscular [48]. All techniques require mobilisation of the ulnar nerve proximal and distal to the medial epicondyle. Efforts should be made to preserve the blood vessels that travel with the nerve. Fine articular branches to the elbow are divided. The first motor branch to FCU can restrain the ulnar nerve, in this situation proximal neurolysis can help with mobility. For subcutaneous transposition a full thickness cutaneous flap including all subdermal adipose is elevated anterior to the medial epicondyle to create a “pocket” for the nerve. The medial intramuscular septum that can be felt as a sharp band on the ridge of the medial humeral condyle, must be excised with care taken to diathermy the vessels that are found at the distal extent to prevent haematoma. The ulnar nerve is then transposed anteriorly into this pocket and the flap is sutured to the fascia anterior to the medial epicondyle to prevent recurrent subluxation. Care is taken to ensure that there is no kinking of the nerve and that the flap is not sutured so tightly so as to compress the nerve. Alternatively the superficial fascia of the flexor-pronator mass can be raised from distal to proximal and the nerve placed between the fascia and muscle mass to create a subfascial transposition, [48] or through the muscle mass by sharp division of septa within the muscle for an intramuscular transposition [49], [50], or lastly deep to the flexor-pronator mass by a technique of ‘Z’ lengthening of the common flexor origin [51].
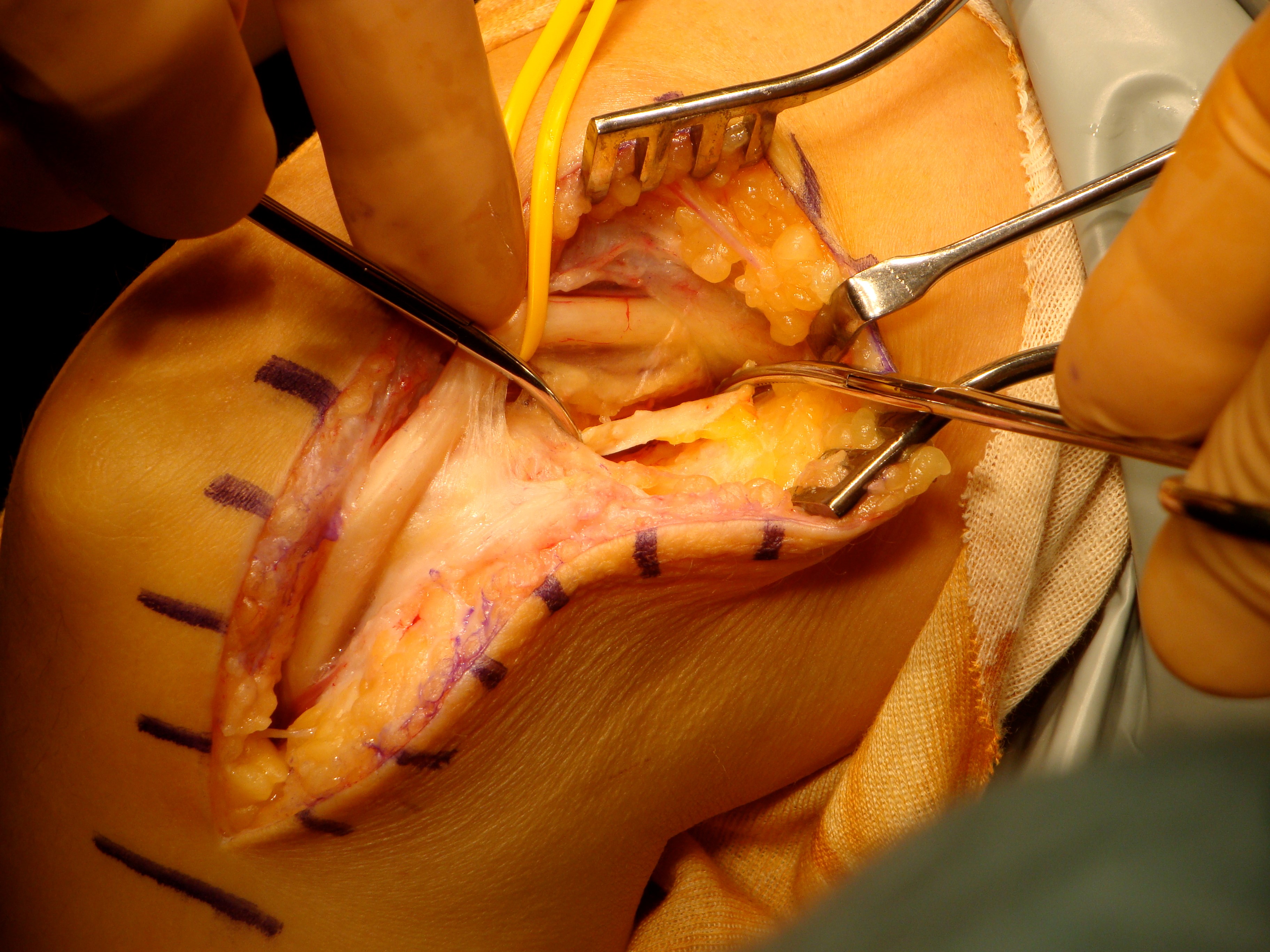
Medial epicondylectomy
Medial epicondylectomy is a treatment alternative, either as a minimal resection, partial resection or total medial epicondylectomy. Total medial epicondylectomy requires the flexor-pronator mass is elevated off the medial epicondyle which is then removed flush with the medial border of the trochlea, and the flexor pronator mass resutured over the raw cancellous surface [52]. This carries the clear risk of injury to the humeral attachment of the medial collateral ligament (MCL) [53]. Unpublished data from our own laboratory has shown that even with subtotal medial epicondylectomy the origin of the MCL may be at risk as there is substantial variation in the site of origin from the medial epicondyle. In an attempt to address this issue other authors have proposed excision of only the posterior portion of the medial epicondyle to preserve the anterior structures [54], [55].
No clear benefit has been found with any one procedure. Comparative studies of in-situ decompression and anterior transposition have found no significant differences in outcome [56], [57], [58], [59], and this is supported by meta-analysis of available evidence [60]. While the reported outcomes appear to be comparable, the complication rate has been found to be higher with anterior transposition techniques [61], [62], [63]. In an attempt to address the shortcomings of published randomised trials and meta-analyses Brauer and Graham performed a decision analysis model that evaluated the balance of risks and benefits of all treatment modalities and concluded that in-situ decompression had the highest utility [64].
There is some debate in the literature about the need for transposition of an unstable nerve [41]. Those in favour argue that the nerve is subject to “friction” as it subluxates anteriorly around the medial epicondyle. There is little evidence to support this supposition however. Childress examined 2,000 elbows and reported that 324 were found to have instability of the nerve, none were symptomatic [20]. Successful outcomes have been reported with in-situ decompression in the presence of an unstable nerve [42].
Author’s preferred surgical technique
It is the author’s preference to perform in-situ decompression in all primary cases without evidence of cubitus valgus or lesion in the floor of the cubital tunnel. The patient is positioned in the lateral decubitus position with a pneumatic tourniquet on the arm. A 4 cm incision is made 1 cm posterior to the medial epicondyle. Blunt dissection is performed to the level of the fascia to protect the posterior branch of the medial cutaneous nerve of the forearm. A subcutaneous tunnel is made on the fascia 7 cm proximally and distally to allow visualisation for decompression. The cutaneous nerves will be elevated with the subdermal fat to which they are adherent. The use of a 5 cm Langenbeck retractor to elevate the tissues will facilitate safe dissection under direct vision. The ulnar nerve is identified and overlying fascia is incised just proximal to the cubital retinaculum. The overlying fascia is then incised under direct vision 7 cm proximally taking care to avoid injury to blood vessels supplying the nerve. A blunt dissector is inserted further to ensure there is no further proximal entrapment. The cubital retinaculum is then incised to the leading edge of FCU where a three-layered decompression is performed. First the FCU fascia is incised sharply, then the FCU muscle is separated in the line of the fibres using blunt dissection to minimise bleeding then the deep (Osborne’s) fascia is incised sharply with dissecting scissors for at least 7 cm distal to the medial epicondyle. Evidence of compression of the nerve is documented. Careful haemostasis is achieved with bipolar diathermy and the elbow is passively flexed to ensure that the nerve remains in the bed. The wound is closed with subcuticular absorbable sutures and early active mobilisation is encouraged.
Expected outcomes
McGowan, in describing her grading system for cubital tunnel syndrome in 1950, reported that for those patients classified Grade 1 with isolated sensory symptoms all had complete resolution following anterior transposition [22]. For those she classified as intermediate (Grade II), with observable muscle weakness, twenty-two of twenty-seven had improvement in symptoms following anterior transposition. Sensory symptoms improved more frequently than motor function. For those with severe lesions with total paresis of at least one ulnar innervated muscle (Grade III) return of normal sensation was observed in 10 out of 13 patients but motor function did not recover so well with only four reporting that the hand felt normal after the surgery. It has been shown however that at long-term follow-up beyond two years after surgery recovery of nerve function can be observed in patients with McGowan grade III lesions [65].
Mowlavi’s meta-analysis of 30 clinical studies suggests that success after any surgical procedure is partly dependent on the preoperative stage of compressive ulnar neuropathy [66]. A number of factors, some unrelated to the neuropathy itself, have been correlated with poorer prognoses. They include age older than 50 years; endocrine comorbidities, especially diabetes mellitus; neuropathies involving multiple peripheral nerves; and chronic neuropathy accompanied by muscle atrophy and poor sensibility.
Nathan et al. reported that nearly 80% of patients had a good or excellent outcome in the long term after in-situ decompression [67]. Using linear regression superior outcomes could be predicted in males, and a worse outcome in those with subluxation of the nerve. Severity of changes on nerve conduction studies did not appear to be predictive of outcome in this study but absent sensory conduction has been shown to be associated with a worse outcome by others [68]. Similar results have been found when studying the long term outcome of epicondylectomy for cubital tunnel syndrome with female gender, ipsilateral carpal tunnel syndrome, patients in their fourth decade and failure to return to work within three months of surgery predictive of recurrence [69].
In young active military personnel the functional recovery has been shown to be good with statistically significant improvement in the average DASH score from over 30 to 6/100 at a minimum of 12 months after surgery [70].
Rehabilitation
Immediate active mobilisation is preferred in our practice. Black et al. found that patients who were immediately mobilised post-surgery returned to work some twenty days earlier than those who went through a three week period of casting following anterior subcutaneous transposition [71]. Weirich compared immediate and delayed mobilisation following subcutaneous transposition and found no significant difference in pain relief, weakness or patient satisfaction [72]. The time period to return to work and normal activities was significantly lower in the immediate mobilisation group.
Complications
The potential complications of treating cubital tunnel syndrome vary according to the surgical technique employed. Delay in presentation, diagnosis or treatment may allow irreversible changes to occur to the nerve. Common to all surgical interventions is the risk of cutaneous nerve injury resulting in numbness around the elbow, scar tenderness or neuroma. For medial epicondylectomy the greatest concern is that the elbow becomes unstable due to overzealous epicondylectomy that defunctions the medial collateral ligament [73]. One author has suggested that inadequate excision is in fact a more common error and that this is associated with a worse outcome [74]. After transposition procedures the most common cause of failure or recurrence is to resect the medial intramuscular septum or to perform an inadequate release distally or proximally. Submuscular transposition may be associated with flexion contracture of the elbow in 5 to 10% of cases although this rarely exceeds 30º [75]. Dense scarring of the nerve may occur with intramuscular transpositions.
Special considerations
Symptomatic patients with normal nerve conduction studies have been shown to do well after cubital tunnel decompression. Tomaino et al., 2001 showed that patients with McGowan type I symptoms and normal nerve conduction studies treated with an in-situ decompression and medial epicondylectomy all found resolution of their paraesthesia and return of grip strength to normal [76].
Treatment of recurrent symptoms may be a challenge. The first objective is to establish that the patient has true recurrence rather than persistence of symptoms, which may occur in up to 75% of patients. Recurrence is determined by a clear history of symptom resolution or improvement after surgical intervention followed by a return or deterioration in symptoms. Nerve conduction studies should be performed and may be compared to pre-operative studies if they are performed in the same laboratory. However, Wilson and Krout reported that post-operative electrodiagnostic studies do not always clarify the diagnosis of recurrent or persistent ulnar nerve compression and therefore many prefer to base the decision on deteriorating sensory and motor function [77]. Commonly cited causes of recurrence include scarring around the nerve, inadequate release, failure to excise the medial intermuscular septum with anterior transposition and instability of the nerve. Broudy and Rogers carried out independent studies into failed ulnar nerve surgery. In 90% the cause was found to be failure to release the medial intermuscular septum [78], [79]. It is important to consider compression of nerve roots in the cervical spine, so that despite release of part of the compression there remains some entrapment, which can be part of a double-crush phenomenon [80].
Historically submuscular transposition was recommended for revision cases. This has been shown to reliably relieve pain but recovery of motor function was reported to be variable [79]. Caputo et al. treated 20 patients with a subcutaneous transposition, and found this to be an effective treatment for recurrence, with 75% good or excellent outcome at two years [81].
Authors preferred management
For patients with McGowan grade 1 cubital tunnel syndrome the author aims for symptom control with activity modification, advice and three-month review. Splints are not well tolerated in the author’s experience. For those with symptom progression or McGowan grade 2 or 3 symptoms the author prefers in-situ decompression through a 5cm incision posterior to the medial epicondyle with the patient in the lateral decubitus position under general anaesthesia. Subcutaneous anterior transposition is undertaken if the patient has recurrent symptoms following previous in-situ decompression. Care is taken to differentiate between recurrence and persistent symptoms.
Ulnar nerve compression at the wrist
The anatomy of the distal ulnar tunnel has been studied in detail and the symptoms of ulnar nerve compression at the wrist can be closely related to the anatomy and site of compression [82]. The distal ulnar tunnel lies between the pisiform and the hook of the hamate and extends from the proximal margin of the volar carpal ligament to the proximal fibrous margin of the hypothenar muscles. The roof of the tunnel is made up of the volar carpal ligament, the palmaris brevis muscle, fat and the overlying fascia of the hypothenar muscles depending on the level at which the tunnel is examined The volar carpal ligament is a condensation of distinct connective tissue in continuity with the deep forearm fascia. It forms the most proximal roof of the tunnel. The floor of the tunnel is made up of the flexor digitorum profundus (FDP) tendons, the transverse carpal ligament, the pisohamate and pisometacarpal ligaments and the opponens digiti minimi (ODM). The terms distal ulnar tunnel and Guyon’s canal are often used interchangeably although Guyon described a space at the base of the hypothenar eminence that is at the most distal part of the distal ulnar tunnel.
The distal ulnar tunnel is divided into three zones based upon the pattern of ulnar nerve branching. Zone 1 is the most proximal part of the tunnel and is defined as the area before the ulnar nerve divides into its deep motor and the mainly sensory superficial components. Nerve compression in zone 1 therefore may affect both modalities. Zone 2 is distal to the bifurcation of the ulnar nerve and includes the area around the deep motor branch of the nerve. Compression here results in motor symptoms with weakness of the intrinsic muscles and possibly also the hypothenar muscles. Zone 3 is also distal to the bifurcation of the nerve but includes the area around the superficial branch of the nerve so that compression in this area causes sensory symptoms, usually to the little and ring fingers.
Although the nerve is macroscopically a single structure in zone 1, microscopic examination has demonstrated that in fact it is often made up of two distinct bundles of nerve fascicles; the more dorsal and ulnar fibres subsequently become the deep motor branch while the more palmar and radial fibres go on to form the superficial branch so that is possible for zone 1 compression to selectively or preferentially result in sensory or motor symptoms [82].
Compression in the distal ulnar tunnel may result from space occupying lesions such as ganglia, anomalous muscles, aneursymal or pseudoaneurysmal swellings of the ulnar artery as well as trauma, tumours or repetitive injury. Sensory symptoms relating to the compression of the superficial branch may include paresthesiae or pain affecting the ulnar one and a half digits. Compression of the deep motor branch may result in wasting of the interossei and the hypothenar eminence as well as intrinsic weakness. These may manifest as reduced grip strength, a reduction in manual dexterity or clumsiness.
Compression may be localised to the distal ulnar tunnel where there is a suggestive history and examination for ulnar nerve compression with normal sensation at the dorsoulnar aspect of the hand and wrist, the absence of localising signs to suggest compression around the elbow or more proximally. Electrodiagnostic testing is useful to confirm the site of compression and local pathology that might be responsible should be actively sought using ultrasound or magnetic resonance imaging (MRI). Computed tomography (CT) will demonstrate a fracture of the hook of hamate. Where this is the case, surgical excision combined with decompression may be curative.
Management for ulnar nerve compression at the wrist
Non-operative treatments may include activity modification and analgesia. Where symptoms are mild or infrequent or the patient has very low functional demands, these techniques may be successful. Often though, persistent or progressive symptoms require escalation of treatment to involve surgery. The natural history of distal ulnar nerve compression is not well documented, partly because of the range of causative factors and an increasing trend to early surgery.
Surgical decompression of the distal ulnar tunnel is a straightforward procedure that can be performed under local anaesthesia, but requires familiarity with the local anatomy in order to achieve a safe and complete release. A tourniquet is not necessary if local anaesthetic with adrenaline is used. It can be achieved using a curved incision similar to that used for carpal tunnel decompression. This runs half a centimetre parallel and ulnar to the thenar crease. The superficial soft tissues are dissected to the volar carpal ligament that is divided longitudinally. Occasionally a communicating cutaneous branch of the ulnar nerve is found in the distal half of the surgical incision and should be protected. The hook of the hamate is palpated and the deep motor branch of the nerve is explored and decompressed along its length splitting the hypothenar muscles, the overlying fascia and releasing any intramuscular tendinous adhesions. The deep branch is most reliably identified at the proximal edge of the hypothenar musculature where it may be found curving around the hook of the hamate. Attention to the release of this ‘fibrous arcade’ prevents residual compression of the deep motor branch [83].
Results of treatment
The results for ulnar nerve decompression at the wrist are not as well reported in the literature as are the results of cubital tunnel surgery. The overall impression is favourable and there is very little to support a non-operative strategy, however there are a number of potential complications after surgery. These include persistent numbness, deep motor nerve injury, vascular injury, infection and wound dehiscence as well as scar and palmar sensitivity.
References
[1] Polatsch DB, Melone CP Jr, Beldner S, Incorvaia A. Ulnar nerve anatomy. Hand Clin. 2007 Aug;23(3):283-9. DOI: 10.1016/j.hcl.2007.05.001[2] Bartels RH, Grotenhuis JA, Kauer JM. The arcade of Struthers: an anatomical study. Acta Neurochir (Wien). 2003 Apr;145(4):295-300; discussion 300. DOI: 10.1007/s00701-003-0006-5
[3] Siqueira MG, Martins RS. The controversial arcade of Struthers. Surg Neurol. 2005;64 Suppl 1:S1:17-20; discussion S1:20-1. DOI: 10.1016/j.surneu.2005.04.017
[4] al-Qattan MM, Murray KA. The arcade of Struthers: an anatomical study. J Hand Surg Br. 1991 Aug;16(3):311-4. DOI: 10.1016/0266-7681(91)90059-W
[5] von Schroeder HP, Scheker LR. Redefining the "Arcade of Struthers". J Hand Surg Am. 2003 Nov;28(6):1018-21. DOI: 10.1016/S0363-5023(03)00421-0
[6] O'Driscoll SW, Horii E, Carmichael SW, Morrey BF. The cubital tunnel and ulnar neuropathy. J Bone Joint Surg Br. 1991 Jul;73(4):613-7. DOI: 10.1302/0301-620X.73B4.2071645
[7] Siemionow M, Agaoglu G, Hoffmann R. Anatomic characteristics of a fascia and its bands overlying the ulnar nerve in the proximal forearm: a cadaver study. J Hand Surg Eur Vol. 2007 Jun;32(3):302-7. DOI: 10.1016/J.JHSB.2006.12.015
[8] Lowe JB 3rd, Maggi SP, Mackinnon SE. The position of crossing branches of the medial antebrachial cutaneous nerve during cubital tunnel surgery in humans. Plast Reconstr Surg. 2004 Sep;114(3):692-6. DOI: 10.1097/01.PRS.0000130966.16460.3C
[9] Vanderpool DW, Chalmers J, Lamb DW, Whiston TB. Peripheral compression lesions of the ulnar nerve. J Bone Joint Surg Br. 1968 Nov;50(4):792-803. DOI: 10.1302/0301-620X.50B4.792
[10] Macnicol MF. Extraneural pressures affecting the ulnar nerve at the elbow. Hand. 1982 Feb;14(1):5-11. DOI: 10.1016/S0072-968X(82)80030-2
[11] Green JR Jr, Rayan GM. The cubital tunnel: anatomic, histologic, and biomechanical study. J Shoulder Elbow Surg. 1999 Sep-Oct;8(5):466-70. DOI: 10.1016/S1058-2746(99)90078-2
[12] Dellon AL, Chang E, Coert JH, Campbell KR. Intraneural ulnar nerve pressure changes related to operative techniques for cubital tunnel decompression. J Hand Surg Am. 1994 Nov;19(6):923-30. DOI: 10.1016/0363-5023(94)90091-4
[13] Iba K, Wada T, Aoki M, Tsuji H, Oda T, Yamashita T. Intraoperative measurement of pressure adjacent to the ulnar nerve in patients with cubital tunnel syndrome. J Hand Surg Am. 2006 Apr;31(4):553-8. DOI: 10.1016/j.jhsa.2006.01.009
[14] Iba K, Wada T, Aoki M, Oda T, Ozasa Y, Yamashita T. The relationship between the pressure adjacent to the ulnar nerve and the disease causing cubital tunnel syndrome. J Shoulder Elbow Surg. 2008 Jul-Aug;17(4):585-8. DOI: 10.1016/j.jse.2007.12.003
[15] Toby EB, Hanesworth D. Ulnar nerve strains at the elbow. J Hand Surg Am. 1998 Nov;23(6):992-7. DOI: 10.1016/S0363-5023(98)80005-1
[16] Kwan MK, Wall EJ, Massie J, Garfin SR. Strain, stress and stretch of peripheral nerve. Rabbit experiments in vitro and in vivo. Acta Orthop Scand. 1992 Jun;63(3):267-72. DOI: 10.3109/17453679209154780
[17] Schuind FA, Goldschmidt D, Bastin C, Burny F. A biomechanical study of the ulnar nerve at the elbow. J Hand Surg Br. 1995 Oct;20(5):623-7. DOI: 10.1016/S0266-7681(05)80124-X
[18] Wall EJ, Massie JB, Kwan MK, Rydevik BL, Myers RR, Garfin SR. Experimental stretch neuropathy. Changes in nerve conduction under tension. J Bone Joint Surg Br. 1992 Jan;74(1):126-9. DOI: 10.1302/0301-620X.74B1.1732240
[19] Gelberman RH, Yamaguchi K, Hollstien SB, Winn SS, Heidenreich FP Jr, Bindra RR, Hsieh P, Silva MJ. Changes in interstitial pressure and cross-sectional area of the cubital tunnel and of the ulnar nerve with flexion of the elbow. An experimental study in human cadavera. J Bone Joint Surg Am. 1998 Apr;80(4):492-501. DOI: 10.2106/00004623-199804000-00005
[20] Childress HM. Recurrent ulnar-nerve dislocation at the elbow. Clin Orthop Relat Res. 1975 May;(108):168-73. DOI: 10.1097/00003086-197505000-00027
[21] Macnicol MF. The results of operation for ulnar neuritis. J Bone Joint Surg Br. 1979 May;61-B(2):159-64. DOI: 10.1302/0301-620X.61B2.438266
[22] McGowan AJ. The results of transposition of the ulnar nerve for traumatic ulnar neuritis. J Bone Joint Surg Br. 1950 Aug;32-B(3):293-301. DOI: 10.1302/0301-620X.32B3.293
[23] Dellon AL, Hament W, Gittelshon A. Nonoperative management of cubital tunnel syndrome: an 8-year prospective study. Neurology. 1993 Sep;43(9):1673-7. DOI: 10.1212/WNL.43.9.1673
[24] Latinovic R, Gulliford MC, Hughes RA. Incidence of common compressive neuropathies in primary care. J Neurol Neurosurg Psychiatry. 2006 Feb;77(2):263-5. DOI: 10.1136/jnnp.2005.066696
[25] Aoki M, Takasaki H, Muraki T, Uchiyama E, Murakami G, Yamashita T. Strain on the ulnar nerve at the elbow and wrist during throwing motion. J Bone Joint Surg Am. 2005 Nov;87(11):2508-14. DOI: 10.2106/00004623-200511000-00019
[26] Rayan GM. Proximal ulnar nerve compression. Cubital tunnel syndrome. Hand Clin. 1992 May;8(2):325-36.
[27] Buehler MJ, Thayer DT. The elbow flexion test. A clinical test for the cubital tunnel syndrome. Clin Orthop Relat Res. 1988 Aug;(233):213-6.
[28] Novak CB, Lee GW, Mackinnon SE, Lay L. Provocative testing for cubital tunnel syndrome. J Hand Surg Am. 1994 Sep;19(5):817-20. DOI: 10.1016/0363-5023(94)90193-7
[29] Kothari MJ, Heistand M, Rutkove SB. Three ulnar nerve conduction studies in patients with ulnar neuropathy at the elbow. Arch Phys Med Rehabil. 1998 Jan;79(1):87-9. DOI: 10.1016/S0003-9993(98)90214-3
[30] Bhala RP. Electrodiagnosis of ulnar nerve lesions at the elbow. Arch Phys Med Rehabil. 1976 May;57(5):206-12.
[31] Tackmann W, Vogel P, Kaeser HE, Ettlin T. Sensitivity and localizing significance of motor and sensory electroneurographic parameters in the diagnosis of ulnar nerve lesions at the elbow. A reappraisal. J Neurol. 1984;231(4):204-11. DOI: 10.1007/BF00313940
[32] Padua L, Aprile I, Mazza O, Padua R, Pietracci E, Caliandro P, Pauri F, D'Amico P, Tonali P. Neurophysiological classification of ulnar entrapment across the elbow. Neurol Sci. 2001 Feb;22(1):11-6. DOI: 10.1007/s100720170030
[33] Caliandro P, Foschini M, Pazzaglia C, La Torre G, Aprile I, Granata G, Tonali P, Padua L. IN-RATIO: a new test to increase diagnostic sensitivity in ulnar nerve entrapment at elbow. Clin Neurophysiol. 2008 Jul;119(7):1600-6. DOI: 10.1016/j.clinph.2008.03.007
[34] Wiesler ER, Chloros GD, Cartwright MS, Shin HW, Walker FO. Ultrasound in the diagnosis of ulnar neuropathy at the cubital tunnel. J Hand Surg Am. 2006 Sep;31(7):1088-93. DOI: 10.1016/j.jhsa.2006.06.007
[35] Britz GW, Haynor DR, Kuntz C, Goodkin R, Gitter A, Maravilla K, Kliot M. Ulnar nerve entrapment at the elbow: correlation of magnetic resonance imaging, clinical, electrodiagnostic, and intraoperative findings. Neurosurgery. 1996 Mar;38(3):458-65; discussion 465. DOI: 10.1097/00006123-199603000-00007
[36] Padua L, Aprile I, Caliandro P, Foschini M, Mazza S, Tonali P. Natural history of ulnar entrapment at elbow. Clin Neurophysiol. 2002 Dec;113(12):1980-4. DOI: 10.1016/S1388-2457(02)00295-X
[37] Seror P. Treatment of ulnar nerve palsy at the elbow with a night splint. J Bone Joint Surg Br. 1993 Mar;75(2):322-7. DOI: 10.1302/0301-620X.75B2.8444959
[38] Svernlöv B, Larsson M, Rehn K, Adolfsson L. Conservative treatment of the cubital tunnel syndrome. J Hand Surg Eur Vol. 2009 Apr;34(2):201-7. DOI: 10.1177/1753193408098480
[39] Dellon AL. Review of treatment results for ulnar nerve entrapment at the elbow. J Hand Surg Am. 1989 Jul;14(4):688-700. DOI: 10.1016/0363-5023(89)90192-5
[40] Goldfarb CA, Sutter MM, Martens EJ, Manske PR. Incidence of re-operation and subjective outcome following in situ decompression of the ulnar nerve at the cubital tunnel. J Hand Surg Eur Vol. 2009 Jun;34(3):379-83. DOI: 10.1177/1753193408101467
[41] Waugh RP, Zlotolow DA. In situ decompression of the ulnar nerve at the cubital tunnel. Hand Clin. 2007 Aug;23(3):319-27, vi. DOI: 10.1016/j.hcl.2007.06.001
[42] Taniguchi Y, Takami M, Takami T, Yoshida M. Simple decompression with small skin incision for cubital tunnel syndrome. J Hand Surg Br. 2002 Dec;27(6):559-62. DOI: 10.1054/jhsb.2002.0821
[43] Bain GI, Bajhau A. Endoscopic release of the ulnar nerve at the elbow using the Agee device: a cadaveric study. Arthroscopy. 2005 Jun;21(6):691-5. DOI: 10.1016/j.arthro.2005.02.018
[44] Hoffmann R, Siemionow M. The endoscopic management of cubital tunnel syndrome. J Hand Surg Br. 2006 Feb;31(1):23-9. DOI: 10.1016/j.jhsb.2005.08.008
[45] Ahcan U, Zorman P. Endoscopic decompression of the ulnar nerve at the elbow. J Hand Surg Am. 2007 Oct;32(8):1171-6. DOI: 10.1016/j.jhsa.2007.07.004
[46] Tsai TM, Bonczar M, Tsuruta T, Syed SA. A new operative technique: cubital tunnel decompression with endoscopic assistance. Hand Clin. 1995 Feb;11(1):71-80.
[47] Watts AC, Bain GI. Patient-rated outcome of ulnar nerve decompression: a comparison of endoscopic and open in situ decompression. J Hand Surg Am. 2009 Oct;34(8):1492-8. DOI: 10.1016/j.jhsa.2009.05.014
[48] Teoh LC, Yong FC, Tan SH, Andrew Chin YH. Anterior subfascial transposition of the ulnar nerve. J Hand Surg Br. 2003 Feb;28(1):73-6. DOI: 10.1054/JHSB.2002.0867
[49] Glowacki KA, Weiss AP. Anterior intramuscular transposition of the ulnar nerve for cubital tunnel syndrome. J Shoulder Elbow Surg. 1997 Mar-Apr;6(2):89-96. DOI: 10.1016/S1058-2746(97)90028-8
[50] Leone J, Bhandari M, Thoma A. Anterior intramuscular transposition with ulnar nerve decompression at the elbow. Clin Orthop Relat Res. 2001 Jun;(387):132-9. DOI: 10.1097/00003086-200106000-00018
[51] Pasque CB, Rayan GM. Anterior submuscular transposition of the ulnar nerve for cubital tunnel syndrome. J Hand Surg Br. 1995 Aug;20(4):447-53. DOI: 10.1016/S0266-7681(05)80151-2
[52] Craven PR Jr, Green DP. Cubital tunnel syndrome. Treatment by medial epicondylectomy. J Bone Joint Surg Am. 1980 Sep;62(6):986-9. DOI:10.2106/00004623-198062060-00016
[53] Tada H, Hirayama T, Katsuki M, Habaguchi T. Long term results using a modified King's method for cubital tunnel syndrome. Clin Orthop Relat Res. 1997 Mar;(336):107-10. DOI: 10.1097/00003086-199703000-00014
[54] Popa M, Dubert T. Treatment of cubital tunnel syndrome by frontal partial medial epicondylectomy. A retrospective series of 55 cases. J Hand Surg Br. 2004 Dec;29(6):563-7. DOI: 10.1016/j.jhsb.2004.06.014
[55] Kaempffe FA, Farbach J. A modified surgical procedure for cubital tunnel syndrome: partial medial epicondylectomy. J Hand Surg Am. 1998 May;23(3):492-9. DOI: 10.1016/S0363-5023(05)80467-8
[56] Bimmler D, Meyer VE. Surgical treatment of the ulnar nerve entrapment neuropathy: submuscular anterior transposition or simple decompression of the ulnar nerve? Long-term results in 79 cases. Ann Chir Main Memb Super. 1996;15(3):148-57. DOI: 10.1016/S0753-9053(96)80004-4
[57] Chan RC, Paine KW, Varughese G. Ulnar neuropathy at the elbow: comparison of simple decompression and anterior transposition. Neurosurgery. 1980 Dec;7(6):545-50. DOI: 10.1227/00006123-198012000-00001
[58] Davies MA, Vonau M, Blum PW, Kwok BC, Matheson JM, Stening WA. Results of ulnar neuropathy at the elbow treated by decompression or anterior transposition. Aust N Z J Surg. 1991 Dec;61(12):929-34. DOI: 10.1111/j.1445-2197.1991.tb00011.x
[59] Nabhan A, Ahlhelm F, Kelm J, Reith W, Schwerdtfeger K, Steudel WI. Simple decompression or subcutaneous anterior transposition of the ulnar nerve for cubital tunnel syndrome. J Hand Surg Br. 2005 Oct;30(5):521-4. DOI: 10.1016/j.jhsb.2005.05.011
[60] Zlowodzki M, Chan S, Bhandari M, Kalliainen L, Schubert W. Anterior transposition compared with simple decompression for treatment of cubital tunnel syndrome. A meta-analysis of randomized, controlled trials. J Bone Joint Surg Am. 2007 Dec;89(12):2591-8. DOI: 10.2106/JBJS.G.00183
[61] Bartels RH, Verhagen WI, van der Wilt GJ, Meulstee J, van Rossum LG, Grotenhuis JA. Prospective randomized controlled study comparing simple decompression versus anterior subcutaneous transposition for idiopathic neuropathy of the ulnar nerve at the elbow: Part 1. Neurosurgery. 2005 Mar;56(3):522-30; discussion 522-30. DOI: 10.1227/01.NEU.0000154131.01167.03
[62] Biggs M, Curtis JA. Randomized, prospective study comparing ulnar neurolysis in situ with submuscular transposition. Neurosurgery. 2006 Feb;58(2):296-304; discussion 296-304. DOI: 10.1227/01.NEU.0000194847.04143.A1
[63] Gervasio O, Gambardella G, Zaccone C, Branca D. Simple decompression versus anterior submuscular transposition of the ulnar nerve in severe cubital tunnel syndrome: a prospective randomized study. Neurosurgery. 2005;56(1):108-17; discussion 117. DOI: 10.1227/01.NEU.0000145854.38234.81
[64] Brauer CA, Graham B. The surgical treatment of cubital tunnel syndrome: a decision analysis. J Hand Surg Eur Vol. 2007 Dec;32(6):654-62. DOI: 10.1016/J.JHSE.2007.07.001
[65] Matsuzaki H, Yoshizu T, Maki Y, Tsubokawa N, Yamamoto Y, Toishi S. Long-term clinical and neurologic recovery in the hand after surgery for severe cubital tunnel syndrome. J Hand Surg Am. 2004 May;29(3):373-8. DOI: 10.1016/j.jhsa.2004.01.010
[66] Mowlavi A, Andrews K, Lille S, Verhulst S, Zook EG, Milner S. The management of cubital tunnel syndrome: a meta-analysis of clinical studies. Plast Reconstr Surg. 2000 Aug;106(2):327-34. DOI: 10.1097/00006534-200008000-00014
[67] Nathan PA, Keniston RC, Meadows KD. Outcome study of ulnar nerve compression at the elbow treated with simple decompression and an early programme of physical therapy. J Hand Surg Br. 1995 Oct;20(5):628-37. DOI: 10.1016/S0266-7681(05)80125-1
[68] Taha A, Galarza M, Zuccarello M, Taha J. Outcomes of cubital tunnel surgery among patients with absent sensory nerve conduction. Neurosurgery. 2004 Apr;54(4):891-5; discussion 895-6. DOI: 10.1227/01.NEU.0000115152.78918.61
[69] Seradge H, Owen W. Cubital tunnel release with medial epicondylectomy factors influencing the outcome. J Hand Surg Am. 1998 May;23(3):483-91. DOI: 10.1016/S0363-5023(05)80466-6
[70] Fitzgerald BT, Dao KD, Shin AY. Functional outcomes in young, active duty, military personnel after submuscular ulnar nerve transposition. J Hand Surg Am. 2004 Jul;29(4):619-24. DOI: 10.1016/j.jhsa.2004.04.011
[71] Black BT, Barron OA, Townsend PF, Glickel SZ, Eaton RG. Stabilized subcutaneous ulnar nerve transposition with immediate range of motion. Long-term follow-up. J Bone Joint Surg Am. 2000 Nov;82-A(11):1544-51. DOI: 10.2106/00004623-200011000-00005
[72] Weirich SD, Gelberman RH, Best SA, Abrahamsson SO, Furcolo DC, Lins RE. Rehabilitation after subcutaneous transposition of the ulnar nerve: immediate versus delayed mobilization. J Shoulder Elbow Surg. 1998 May-Jun;7(3):244-9. DOI: 10.1016/S1058-2746(98)90052-0
[73] Froimson AI, Anouchi YS, Seitz WH Jr, Winsberg DD. Ulnar nerve decompression with medial epicondylectomy for neuropathy at the elbow. Clin Orthop Relat Res. 1991 Apr;(265):200-6.
[74] Heithoff SJ. Cubital tunnel syndrome does not require transposition of the ulnar nerve. J Hand Surg Am. 1999 Sep;24(5):898-905. DOI: 10.1053/jhsu.1999.0898
[75] Gabel GT, Amadio PC. Reoperation for failed decompression of the ulnar nerve in the region of the elbow. J Bone Joint Surg Am. 1990 Feb;72(2):213-9. DOI: 10.2106/00004623-199072020-00008
[76] Tomaino MM, Brach PJ, Vansickle DP. The rationale for and efficacy of surgical intervention for electrodiagnostic-negative cubital tunnel syndrome. J Hand Surg Am. 2001 Nov;26(6):1077-81. DOI: 10.1053/jhsu.2001.26327
[77] Wilson DH, Krout R. Surgery of ulnar neuropathy at the elbow: 16 cases treated by decompression without transposition. Technical note. J Neurosurg. 1973 Jun;38(6):780-5. DOI: 10.3171/jns.1973.38.6.0780
[78] Broudy AS, Leffert RD, Smith RJ. Technical problems with ulnar nerve transposition at the elbow: findings and results of reoperation. J Hand Surg Am. 1978 Jan;3(1):85-9. DOI: 10.1016/S0363-5023(78)80121-X
[79] Rogers MR, Bergfield TG, Aulicino PL. The failed ulnar nerve transposition. Etiology and treatment. Clin Orthop Relat Res. 1991 Aug;(269):193-200.
[80] Upton AR, McComas AJ. The double crush in nerve entrapment syndromes. Lancet. 1973 Aug 18;2(7825):359-62. DOI: 10.1016/S0140-6736(73)93196-6
[81] Caputo AE, Watson HK. Subcutaneous anterior transposition of the ulnar nerve for failed decompression of cubital tunnel syndrome. J Hand Surg Am. 2000 May;25(3):544-51. DOI: 10.1053/jhsu.2000.6005
[82] Gross MS, Gelberman RH. The anatomy of the distal ulnar tunnel. Clin Orthop Relat Res. 1985 Jun;(196):238-47.
[83] Ombaba J, Kuo M, Rayan G. Anatomy of the ulnar tunnel and the influence of wrist motion on its morphology. J Hand Surg Am. 2010 May;35(5):760-8. DOI: 10.1016/j.jhsa.2010.02.028



