[Prävalenz von HIV bei Beschäftigten im Gesundheitswesen in der Post-HAART-Ära – eine systematische Übersicht und Metaanalyse]
Roland Diel 1Albert Nienhaus 2,3
1 Institute of Epidemiology, University Medical Hospital Schleswig-Holstein, Kiel, Germany
2 Institution for Statutory Accident Insurance and Prevention in the Health and Welfare Services (BGW), Hamburg, Germany
3 Competence Center for Epidemiology and Health Services Research for Healthcare Professionals (CVcare), Institute for Health Services Research in Dermatology and Nursing (IVDP), University Medical Center Hamburg-Eppendorf (UKE), Hamburg, Germany
Zusammenfassung
Hintergrund: Medizinisches Personal (MP) ist potenziell dem Risiko einer HIV-Infektion durch berufliche Exposition ausgesetzt. Mit der Einführung der hochaktiven antiretroviralen Therapie (HAART), der konsequenten Anwendung von Basishygienemaßnahmen sowie der Verfügbarkeit einer Postexpositionsprophylaxe hat sich das globale Risikoprofil jedoch grundlegend verändert.
Ziel: Ziel dieser Studie war es, weltweit Daten zur HIV-Prävalenz bei MP zu erheben, um aktuelle epidemiologische Muster zu erfassen und regionale Unterschiede zu bewerten.
Methode: In Übereinstimmung mit den PRISMA-Leitlinien wurde eine systematische Literaturrecherche in PubMed, Scopus und der Cochrane Library durchgeführt. Eingeschlossen wurden Studien, die zwischen 1996 und September 2025 – entsprechend der Post-HAART-Ära – über serologische Nachweise von HIV-Infektionen bei MP berichteten. Extrahiert wurden Angaben zum Studiendesign, zur Stichprobengröße und zur Anzahl HIV-positiver Fälle. Prävalenzschätzungen wurden mit internen Kontrollgruppen oder mit nationalen Hintergrundprävalenzen verglichen, die aus UNAIDS-Daten gewonnen wurden. Die methodische Qualität der Studien wurde mithilfe des Westermann-Scores bewertet. Zur Schätzung der gepoolten HIV-Prävalenz nach WHO-Regionen wurden Random-Effects-Metaanalysen durchgeführt.
Ergebnisse: Von insgesamt 220 identifizierten Studien erfüllten 14 aus 12 Ländern die Einschlusskriterien. Bei insgesamt 7.705 getesteten Beschäftigten im Gesundheitswesen betrug die gepoolte HIV-Prävalenz 0,68% (95%-KI 0,19–2,46) mit allerdings ausgeprägter Heterogenität (I²=94,3%, τ²=5,003). Während die gepoolte Prävalenz unter MP in subsaharischen Afrika 7,1% (95%-KI 3,3–12,7) betrug, zeigten alle anderen Regionen – Europa, Lateinamerika und der östliche Mittelmeerraum – eine gepoolte Prävalenz von 0,0% (95%-KI 0,0–3,7), wobei keine HIV-positiven Beschäftigten nachgewiesen wurden.
Schlussfolgerungen: HIV-Infektionen bei MP sind weltweit mittlerweile äußerst selten, wobei erhöhte Prävalenzen auf Hochendemiegebiete in Afrika beschränkt sind. Die Ergebnisse sprechen dafür, dass HIV-Infektionen bei MP überwiegend durch Transmissionen in der Allgemeinbevölkerung und nicht durch berufliche Exposition bedingt sind. Gleichzeitig unterstreichen sie den Erfolg von Infektionsschutzmaßnahmen und der breiten Verfügbarkeit antiretroviraler Therapie zur Eliminierung beruflicher HIV-Übertragungen. Eine fortgesetzte Surveillance sowie ein bedarfsgerechter Zugang zu persönlicher Schutzausrüstung und Postexpositionsprophylaxe bleiben jedoch essenziell, um diesen bedeutenden Erfolg im Arbeitsschutz dauerhaft zu sichern.
Schlüsselwörter
HIV, medizinisches Personal, berufliche Exposition, Prävalenz, hochaktive antiretrovirale Therapie, HAART, Metaanalyse
Introduction
Globally, approximately 39 million people were living with HIV in 2023, according to the Joint United Nations Programme on HIV/AIDS (UNAIDS) and the World Health Organization (WHO), corresponding to a global prevalence of about 0.7% among adults aged 15–49 years [1], [2]. The highest burden remains concentrated in sub-Saharan Africa, where prevalence still exceeds 5% in several countries, whereas rates in high-income regions such as Europe and North America have stabilized at below 0.3% [1], [2], [3].
The overall epidemiological landscape of HIV infection has therefore shifted from an expanding global epidemic in the late twentieth century to a controlled chronic infection, particularly in industrialized countries with widespread access to antiretroviral therapy (ART) [4], [5]. Healthcare workers (HCWs), however, represent a unique subpopulation because of their occupational exposure to blood and body fluids. Before the advent of highly active antiretroviral therapy (HAART) in 1996, the potential for occupational HIV transmission was a matter of intense concern. Documented seroconversions following needlestick injuries and surgical accidents in the 1980s and early 1990s led to major anxiety within the healthcare sector and to restrictive national policies that excluded HIV-infected HCWs from performing exposure-prone procedures (EPPs) [6], [7].
Since 1996, the introduction of HAART—particularly triple-combination therapy including protease inhibitors—has fundamentally changed the natural history of HIV infection. Viral suppression achieved through effective ART virtually eliminates the risk of secondary transmission [8], [9]. This scientific breakthrough transformed both clinical management and public-health policy, shifting the focus from exclusion and risk avoidance toward evidence-based inclusion and proportionality. Nevertheless, it remains unclear how these therapeutic advances have translated into real-world epidemiological outcomes among healthcare personnel.
The actual prevalence of HIV infection among HCWs in the post-HAART era has never been systematically synthesized. Such a comprehensive global analysis is crucial to determine to which extent occupational exposure continues to contribute to infection risk and to assess whether any measurable occupational excess persists compared with general-population prevalence. Despite global progress, needlestick and sharps injuries, mucocutaneous contact, and surgical incidents continue to occur even under modern infection-control standards [10], [11], [12]. Understanding how frequently such exposures still result in HIV infection is therefore essential for maintaining an evidence-based and proportionate approach to occupational health and infection prevention.
The present systematic review considers all available studies published since 1996 that report HIV prevalence or serostatus among healthcare workers. By focusing exclusively on the post-HAART period, it seeks to clarify the magnitude of residual occupational risk in the context of effective ART and modern prevention practices. In addition, the review compares HCW data with background population prevalence, placing occupational infection risk within its broader epidemiological and public-health context.
This approach provides the first consolidated global assessment of HIV burden in healthcare personnel in nearly three decades and aims to inform future infection-control policies, risk communication, and occupational-health strategies.
Methods
Definition of HCWs
HCWs were defined as all medical, dental, nursing, obstetric or assisting personnel working in different areas, e.g. hospitals, outpatient clinics, doctors’ practices, dialysis facilities, nursing homes and out-patient care facilities. The decisive factor was the existence of a plausible transmission pathway within these activities.
Literature search and study selection
A comprehensive literature search up to September 1, 2025, was conducted in PubMed, Scopus and the Cochrane library to identify reports describing the baseline prevalence of HIV infection among HCWs. The following Boolean search string was adapted for each database (“HIV” OR “Human Immunodeficiency Virus”) AND (“prevalence” OR “seroprevalence”) AND (“healthcare workers” OR “health care workers” OR “health personnel”) AND “occupational exposure” AND (1996:[Year]–2025[Year]). Search strategies were tailored to the indexing systems and functionalities of each database to maximise both sensitivity and specificity. Full search strings for both databases, together with the methodological rationale for their design, are presented in Attachment 1 [Att. 1].
Studies providing original serological data on HIV infection in a defined healthcare worker population were eligible for inclusion. Studies limited to post-exposure or seroconversion analyses, as well as review articles, guidelines, conference abstracts, commentaries, and editorials, were excluded, along with articles whose central theme diverged from or was unrelated to HIV prevalence. No restrictions were applied regarding language, study design, subpopulation, or data-collection mode (prospective or retrospective). Reference lists of included studies and relevant reviews were manually screened to identify additional eligible publications. All records were managed using EndNote, which automatically removed duplicates. The review followed the PRISMA 2020 guidelines for systematic reviews and meta-analyses [13], [14].
Data extraction
Two reviewers (RD and AN) independently screened titles, abstracts, and full texts, resolving discrepancies by consensus. From each study, data were extracted on the year and country of publication, study design, study period, HIV-testing method, number tested, number HIV-positive, and the presence or absence of a comparison group. Internal control groups were retained where available.
For studies without an internal comparator, HIV prevalence estimates for the corresponding country and year were derived from UNAIDS [15] or other national population-based surveys and surveillance reports, e.g. the Polish National Institute of Public Health – National Institute of Hygiene (NIZP-PIB) [16], representing the general adult population (aged 15–49 years). When studies reported multi-year periods, the UNAIDS estimate for the midpoint year was selected to ensure temporal consistency. To maintain comparability with global HIV surveillance systems, countries were grouped according to the WHO regional classification.
Quantitative synthesis and meta-analysis
A quantitative synthesis was performed to estimate the pooled HIV prevalence among HCWs. Given the substantial and expected heterogeneity in prevalence arising from vastly different epidemiological contexts across countries, the analysis was stratified a priori by geographical region (Sub-Saharan Africa, Europe, North America, Asia, and Latin America).
Within each geographical stratum, the pooled prevalence was estimated using a random-effects model, which accounts for both within-study variance and between-study variance (τ²). Statistical analyses were performed using the metafor package (version 4.4-0) in the R statistical environment (version 4.3.1). Prevalence estimates with 95% confidence intervals for individual studies were calculated using the exact Clopper-Pearson method. Heterogeneity was quantified using the I² statistic.
Assessment of study quality
Study quality was evaluated using the Westermann 9-item checklist which assesses methodological clarity, representativeness, and laboratory reliability [17]. Each fulfilled item scored one point (maximum=9); studies were classified as low (≤4), moderate (5–7), or high (≥8) quality]. For studies with zero HIV-positive participants, a Zero-Event-Adjusted (ZEA) scoring was applied, treating confirmatory testing as not applicable and rescaling totals to a nine-point maximum (×9/8). ZEA scores were first calculated to one decimal place and then rounded up to the next whole-number value for categorical classification. This adjustment prevents artificial downgrading of zero-event studies that could not perform confirmatory testing because no positive results were obtained, thereby maintaining comparability across all studies. Both versions (all items scored 0–1) were applied.
Results
Study availability
Figure 1 [Fig. 1] shows the flow diagram of the literature search. In total, 220 abstracts were identified (137 in PubMed, 82 in Scopus and 1 in the Cochrane Library), with reviews being excluded by default in the search strategy; 135 studies, of those 28 were duplicates, were excluded based on their abstracts. 85 full-text articles were reviewed. Of these, 14 peer-reviewed studies met the eligibility criteria [18], [19], [20],[21], [22], [23], [24], [25], [26], [27], [28], [29], [30], [31] and were included in the analysis.
Figure 1: Prisma flow diagram of study selection
Study design
Thirteen of the 14 studies (92.9%) were cross-sectional investigations. One study (7.1%) was a prospective cohort design where blood was drawn from Dutch expatriate HCWs, on average, 30 days post-return.
Study characteristics
The characteristics of the included studies are summarized in Attachment 2 [Att. 2], Tab. 1 . All 14 studies were published between 1998 and 2025 and came from 12 different countries. Two studies each were conducted in South Africa and Poland (2/14 or 14.3%), while Brazil, Burkina Faso, Cameroon, Denmark, Georgia, Iraq, Mozambique, the Netherlands, Turkey and Pakistan each contributed one (1/14 or 7.1%). Study sample sizes ranged from 99 to 1,386 participants.
The total number of HCWs serologically tested for HIV antibodies across all studies was 7,705, with 179 HIV-positive individuals. The crude (unweighted) aggregated prevalence representing the simple overall proportion of HIV-positive cases was therefore 2.32% (95% CI 2.00–2.68%, Clopper–Pearson exact). The global random-effects pooled prevalence, however, was 0.68%, (95% CI 0.19–2.46%) and reflects the high weighting influence of numerous zero-event studies outside Sub-Saharan Africa and the extreme inter-regional heterogeneity (I²=94.3%, τ²=5.003). Table 1 [Tab. 1] summarizes the regional distribution and corresponding pooled prevalences.
Table 1: Pooled HIV prevalences among healthcare workers, stratified by region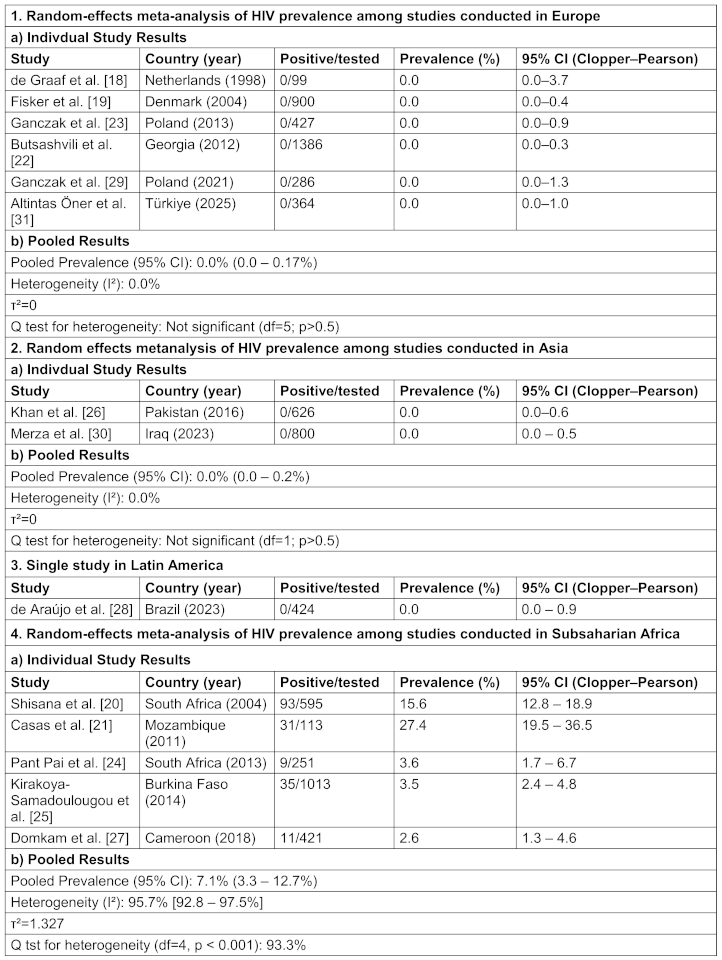
Stratified by WHO regions, all five European studies reported zero HIV-positive cases (0/3462), yielding a pooled prevalence of 0.0% (95% CI: 0.0–0.17%) and no heterogeneity (I²=0%,τ²=0), indicating highly consistent findings across settings in Europe. Similarly, in the Eastern Mediterranean region (Pakistan and Iraq), two cross-sectional studies screened a total of 1 426 HCWs, detecting no HIV-positive cases and yielding a pooled prevalence of 0.0% (95% CI: 0.0–0.2%). No heterogeneity was detected (I²=0%, τ²=0). This consistency across Pakistan and Iraq supports the inference of negligible occupational HIV risk for HCWs also in these low-prevalence settings. Latin American region (Brazil) was represented by a single study from Brazil with no HIV-positive HCW (prevalence of 0.0%; 95% CI: 0.0-0.9), so no meta-analysis was possible.
In contrast, substantial heterogeneity was observed across the five African studies (I²=95.7%), with individual prevalence estimates ranging from 2.6% to 27.4%. The highest proportions originated from southern Africa (South Africa and Mozambique), while studies from West and Central Africa (Burkina Faso, Cameroon) reported considerably lower rates (2–4 %), consistent with background population prevalence levels. The pooled random-effects estimate yielded an overall HIV prevalence of 7.1% (95% CI: 3.3–12.7%), however, the wide confidence interval and the high I² of 95.7% reflect marked variability in prevalence across countries and years. Thus, the pooled estimate for HIV prevalence among African HCWs should be interpreted as an average of widely varying national prevalences rather than a single, true prevalence.
Summary of methodological quality
The methodological quality of the 14 included studies was evaluated using the nine-item Westermann checklist [17]. Under strict scoring, total scores ranged from 4 to 9 points (median=7). Most studies (11/14; 79%%) were rated as moderate quality (5–7 points), one as low quality (=4 points), and two as high quality (=8 points).
To account for studies reporting zero HIV-positive participants, a Zero-Event-Adjusted (ZEA) scoring was applied, in which the confirmatory-test criterion was treated as not applicable, and totals were rescaled to a nine-point maximum (×9/8). ZEA scoring slightly increased scores for zero-event studies (mean +0.6 points), but only one study changed quality category (Attachment 2 [Att. 2]).
Comparison with internal and external control groups
In this review, UNAIDS estimates and other population-based surveillance data were used to determine whether observed HIV prevalence among HCWs aligned with, exceeded, or fell below that of the general adult population during the same period and within the same country. In nearly all cases—with exception of two African datasets, those of Kirakoya-Samadoulougou et al. in Burkina Faso [25] and Casas et al. in Mozambique [21]—HCW prevalence equaled or was lower than national background levels. This pattern indicates that HIV infections among HCWs predominantly reflect community-level epidemiology rather than occupational transmission.
The two outlier studies showing higher HIV prevalence were both substantially affected by selection bias, which likely inflated the observed infection rates. In the study by Casas et al. [21], an extraordinarily high overall HIV prevalence of 43.8% among hospital staff was reported. However, only 47.5% of the workforce participated, almost certainly attracting individuals already aware of their HIV status or perceiving themselves at higher risk. This bias persisted even within the subset of 113 previously untested HCWs, among whom 31 new infections (27.4%) were identified. Because testing was voluntary and non-anonymous, this subgroup was also not representative, and the reported prevalence likely overestimates the true infection rate among the hospital’s total staff. Similarly, the national survey by Kirakoya-Samadoulougou et al. [25] suffered from participation bias. Although the overall prevalence was clearly lower (3.5%), the testing acceptance rate was only 64.5%, and medical doctors constituted merely 1% of participants, rendering the sample non-representative. Comparison with general population data is further complicated by demographic differences: the HCW cohort was older and more urbanized, both independent risk factors for HIV. After statistical adjustment for these confounders, the HIV prevalence among male HCWs (2.5%) became nearly identical to that of men in the general Demographic and Health Surveys (DHS) population (2.3%), demonstrating that the apparent excess risk was attributable to demographic composition rather than occupation. Finally, Fisker et al. [19] provided the only dataset including an internal comparison group of non-HCWs from the same population (0/539; 95% CI 0–0.68%). Their results mirrored the background epidemic level in Denmark, further supporting the absence of occupationally related excess risk.
Discussion
To our knowledge, this analysis represents the first systematic review of published reports on HIV prevalence among HCWs since the introduction of HAART. Across 14 studies with serological test results spanning 28 years (1998–2025), only 179 HIV infections were identified among a total of 7,606 HCWs tested. Although a crude global HIV prevalence of 2.38% suggests that infection among HCWs is rare, this aggregate figure masks profound geographical disparities.
HIV prevalence among HCWs shows a strikingly regional pattern, statistically confirming a pronounced divide. Elevated HCW prevalence is observed only in the context of generalized epidemics in sub-Saharan Africa, where prevalence remains substantial and highly heterogeneous. These figures mirror the broader regional epidemic and reflect the diversity of national HIV contexts. In contrast, in Europe, North America, Asia, and Latin America, HIV prevalence among HCWs is statistically indistinguishable from zero, with pooled estimates consistently at 0% and — depending on study size — extremely narrow upper confidence limits. This provides robust quantitative evidence that occupational HIV transmission risk is negligible in low-prevalence settings.
This interpretation is supported by the landmark study of de Graaf et al. [18], which effectively served as a natural experiment. The study examined a cohort of Dutch expatriate HCWs working in AIDS-endemic countries — individuals with high occupational exposure risk but originating from a low-prevalence population. The finding of zero seroconversions despite frequent needlestick injuries clearly demonstrates that the infection risk profile of the population of origin is a stronger determinant of HIV acquisition than occupational exposure itself.
The pronounced heterogeneity observed within the African stratum (I²>95%) confirms that HCWs cannot be regarded as a uniform risk group. Their infection risk largely mirrors the background HIV prevalence of the communities in which they live and work, rather than occupational exposure. In addition to this statistical heterogeneity, there is a near-complete absence of any measurable occupational excess risk. When benchmarked against UNAIDS or national surveillance estimates, HIV prevalence among HCWs is consistently equal to or lower than that of the general population across all WHO regions. This pattern supports the conclusion that, in the HAART era and under effective infection-control standards, the occupational transmission risk of HIV in healthcare settings is negligible.
The only two apparent outliers — studies from Mozambique [21] and Burkina Faso [25] — reported comparatively elevated HIV prevalence among HCWs, but these findings are likely inflated by methodological bias, particularly self-selection of participants. In the Burkina Faso study, the apparent excess risk among male HCWs disappeared after adjustment for confounders such as age and place of residence, indicating that demographic rather than occupational factors explained the observed differences. These methodological limitations likely account for the higher HCW prevalence compared with the surrounding general population and underscore the importance of representative sampling and independent testing frameworks in occupational-health surveillance. Consequently, these skewed prevalence figures should not be interpreted as representative benchmarks for occupational risk among African HCWs.
In summary, the pronounced geographical dichotomy — where HCW HIV prevalence is virtually zero in low-prevalence regions and rises in parallel with community prevalence in high-burden countries — strongly indicates that community acquisition, not occupational exposure, is the dominant pathway of infection in this professional group. While occupational risk cannot be considered absolutely zero, its contribution to overall infection burden is negligible compared with the influence of background community transmission.
Consistently near-zero prevalence in Europe, the Americas, and Asia underscores the effectiveness of infection-control practices, personal protective equipment (PPE), and post-exposure prophylaxis (PEP). This conclusion is further supported by longitudinal CDC surveillance data [32], which documented a sharp decline in occupationally acquired HIV in the United States: 58 confirmed and 150 possible cases were reported between 1985 and 2013, but only one confirmed case since 1999. The CDC attributed this success to the widespread adoption of standard precautions, engineered sharps protection, and PEP availability in the HAART era.
These findings reinforce the rationale for modern, non-restrictive, U=U-aligned guidelines (“Undetectable=Untransmittable”), such as the UK Advisory Panel for Healthcare Workers Living with Bloodborne Viruses (UKAP) 2024 [33] and the Society for Healthcare Epidemiology of America (SHEA) 2022 [34] recommendations, which permit virally suppressed HCWs to perform all procedures while ensuring equitable access to prevention and treatment resources in under-resourced settings.
Nevertheless, sustained vigilance is warranted — particularly in high-burden African settings — where structural barriers, limited resources, and inconsistent access to protective equipment persist. Strengthening surveillance systems and maintaining investment in infection-control capacity are essential to ensure that the near-elimination of occupational HIV transmission remains a durable public-health achievement.
Conclusions
Together with CDC surveillance data, our findings demonstrate that occupational HIV transmission among HCWs has become exceedingly uncommon since the HAART era and that infection rates largely mirror background epidemics within their respective regions. Maintaining universal precautions, ensuring timely access to PEP, and sustaining early ART coverage remain essential pillars of occupational HIV prevention.
However, the residual heterogeneity observed in sub-Saharan Africa underscores persistent challenges, including resource limitations, incomplete PPE coverage, and a higher community burden. Sustained investment in infection-control infrastructure and continued monitoring of occupational exposures are crucial to consolidate these achievements and ensure the permanent elimination of occupational HIV transmission.
Notes
Author’s ORCID
- Roland Diel: 0000-0001-8304-7709
- Albert Nienhaus: 0000-0003-1881-7302
Competing interests
The authors declare that they have no competing interests.
References
[1] UNAIDS. Global HIV & AIDS statistics — Fact Sheet 2024. Geneva: Joint United Nations Programme on HIV/AIDS (UNAIDS); 2024.[2] World Health Organization (WHO). Global HIV Programme: Data and statistics, 2024 update. Geneva: WHO; 2024.
[3] Centers for Disease Control and Prevention (CDC). Global HIV Surveillance Report, 2023. Atlanta (GA): CDC; 2023.
[4] Mocroft A, Ledergerber B, Katlama C, Kirk O, Reiss P, d'Arminio Monforte A, Knysz B, Dietrich M, Phillips AN, Lundgren JD; EuroSIDA study group. Decline in the AIDS and death rates in the EuroSIDA study: an observational study. Lancet. 2003 Jul 5;362(9377):22-9. DOI: 10.1016/s0140-6736(03)13802-0
[5] Hogg RS, Heath KV, Yip B, Craib KJ, O'Shaughnessy MV, Schechter MT, Montaner JS. Improved survival among HIV-infected individuals following initiation of antiretroviral therapy. JAMA. 1998 Feb 11;279(6):450-4. DOI: 10.1001/jama.279.6.450
[6] Recommendations for preventing transmission of human immunodeficiency virus and hepatitis B virus to patients during exposure-prone invasive procedures. MMWR Recomm Rep. 1991 Jul 12;40(RR-8):1-9.
[7] Department of Health (UK). Guidance for clinical health care workers: protection against infection with blood-borne viruses: recommendations of the Expert Advisory Group on AIDS and the Advisory Group on Hepatitis. London: HMSO; 1991. Available from: https://assets.publishing.service.gov.uk/media/5a7daf95ed915d2ac884cf1e/clinical_health_care_workers_ infection_ bloodborne_viruses.pdf
[8] Rodger AJ, Cambiano V, Bruun T, Vernazza P, Collins S, Degen O, Corbelli GM, Estrada V, Geretti AM, Beloukas A, Raben D, Coll P, Antinori A, Nwokolo N, Rieger A, Prins JM, Blaxhult A, Weber R, Van Eeden A, Brockmeyer NH, Clarke A, Del Romero Guerrero J, Raffi F, Bogner JR, Wandeler G, Gerstoft J, Gutiérrez F, Brinkman K, Kitchen M, Ostergaard L, Leon A, Ristola M, Jessen H, Stellbrink HJ, Phillips AN, Lundgren J; PARTNER Study Group. Risk of HIV transmission through condomless sex in serodifferent gay couples with the HIV-positive partner taking suppressive antiretroviral therapy (PARTNER): final results of a multicentre, prospective, observational study. Lancet. 2019 Jun 15;393(10189):2428-2438. DOI: 10.1016/S0140-6736(19)30418-0
[9] Cohen MS, Chen YQ, McCauley M, Gamble T, Hosseinipour MC, Kumarasamy N, Hakim JG, Kumwenda J, Grinsztejn B, Pilotto JH, Godbole SV, Mehendale S, Chariyalertsak S, Santos BR, Mayer KH, Hoffman IF, Eshleman SH, Piwowar-Manning E, Wang L, Makhema J, Mills LA, de Bruyn G, Sanne I, Eron J, Gallant J, Havlir D, Swindells S, Ribaudo H, Elharrar V, Burns D, Taha TE, Nielsen-Saines K, Celentano D, Essex M, Fleming TR; HPTN 052 Study Team. Prevention of HIV-1 infection with early antiretroviral therapy. N Engl J Med. 2011 Aug 11;365(6):493-505. DOI: 10.1056/NEJMoa1105243
[10] Auta A, Adewuyi EO, Tor-Anyiin A, Edor JP, Kureh GT, Khanal V, Oga E, Adeloye D. Global prevalence of percutaneous injuries among healthcare workers: a systematic review and meta-analysis. Int J Epidemiol. 2018 Dec 1;47(6):1972-1980. DOI: 10.1093/ije/dyy208
[11] Prüss-Üstün A, Rapiti E, Hutin Y. Sharps injuries: Global burden of disease from sharps injuries to health-care workers. Geneva: World Health Organization; 2003. Available from: https://iris.who.int/server/api/core/bitstreams/bd32c4e2-1774-4968-9858-dd8b15a6ea1a/content
[12] Tarantola A, Abiteboul D, Rachline A. Infection risks following accidental exposure to blood or body fluids in health care workers: a review of pathogens transmitted in published cases. Am J Infect Control. 2006 Aug;34(6):367-75. DOI: 10.1016/j.ajic.2004.11.011
[13] Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, Shamseer L, Tetzlaff JM, Akl EA, Brennan SE, Chou R, Glanville J, Grimshaw JM, Hróbjartsson A, Lalu MM, Li T, Loder EW, Mayo-Wilson E, McDonald S, McGuinness LA, Stewart LA, Thomas J, Tricco AC, Welch VA, Whiting P, Moher D. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021 Mar 29;372:n71. DOI: 10.1136/bmj.n71
[14] Munn Z, Moola S, Lisy K, Riitano D, Tufanaru C. Methodological guidance for systematic reviews of observational epidemiological studies reporting prevalence and cumulative incidence data. Int J Evid Based Healthc. 2015 Sep;13(3):147-53. DOI: 10.1097/XEB.0000000000000054
[15] UNAIDS estimates. Joint United Nations Programme on HIV/AIDS (UNAIDS). [date accessed 2025 Aug 27]. Available from: aidsinfo.unaids.org
[16] Werbińska-Sienkiewicz B, Rosińska M, Furman S. Zakazenia HIV i zachorowania na AIDS w Polsce w 2009 roku [HIV and AIDS in Poland in 2009]. Przegl Epidemiol. 2011;65(2):289-95.
[17] Westermann C, Peters C, Lisiak B, Lamberti M, Nienhaus A. The prevalence of hepatitis C among healthcare workers: a systematic review and meta-analysis. Occup Environ Med. 2015 Dec;72(12):880-8. DOI: 10.1136/oemed-2015-102879
[18] de Graaf R, Houweling H, van Zessen G. Occupational risk of HIV infection among western health care professionals posted in AIDS endemic areas. AIDS Care. 1998 Aug;10(4):441-52. DOI: 10.1080/09540129850123975
[19] Fisker N, Mygind LH, Krarup HB, Licht D, Georgsen J, Christensen PB. Blood borne viral infections among Danish health care workers--frequent blood exposure but low prevalence of infection. Eur J Epidemiol. 2004;19(1):61-7. DOI: 10.1023/b:ejep.0000013397.51614.d4
[20] Shisana O, Hall EJ, Maluleke R, Chauveau J, Schwabe C. HIV/AIDS prevalence among South African health workers. S Afr Med J. 2004 Oct;94(10):846-50.
[21] Casas EC, Decroo T, Mahoudo JA, Baltazar JM, Dores CD, Cumba L, De Weggheleire A, Huyst V, Bottieau E. Burden and outcome of HIV infection and other morbidities in health care workers attending an Occupational Health Program at the Provincial Hospital of Tete, Mozambique. Trop Med Int Health. 2011 Nov;16(11):1450-6. DOI: 10.1111/j.1365-3156.2011.02853.x
[22] Butsashvili M, Kamkamidze G, Kajaia M, Morse DL, Triner W, Dehovitz J, et al. Occupational exposure to body fluids among health care workers in Georgia. Occup Med (Lond). 2012 Dec;62(8):620-6. DOI: 10.1093/occmed/kqs121
[23] Gańczak M, Szych Z, Szczeniowski A, Dmytrzyk-Danilów G. Postawy personelu medycznego wobec osób zakazonych HBV, HCV lub HIV pracujacych w oddziałach zabiegowych oraz badania serologiczne pracowników [Attitudes of medical specialists toward HBV, HCV or HIV infected surgical staff and a sero-survey among staff members]. Med Pr. 2013;64(5):639-47. DOI: 10.13075/mp.5893.2013.0060
[24] Pant Pai N, Behlim T, Abrahams L, Vadnais C, Shivkumar S, Pillay S, Binder A, Deli-Houssein R, Engel N, Joseph L, Dheda K. Will an unsupervised self-testing strategy for HIV work in health care workers of South Africa? A cross sectional pilot feasibility study. PLoS One. 2013 Nov 27;8(11):e79772. DOI: 10.1371/journal.pone.0079772
[25] Kirakoya-Samadoulougou F, Fao P, Yaro S, Defer MC, Meda N, Robert A, Nagot N. Are healthcare workers at higher risk of HIV infection than the general population in Burkina Faso? Epidemiol Infect. 2014 Mar;142(3):662-70. DOI: 10.1017/S0950268813001404
[26] Khan MZ, Saqib S, Gardyzi SIHS, Qazi J. Prevalence of blood-borne viruses in health care workers of a northern district in Pakistan: Risk factors and preventive behaviors. Can J Infect Dis Med Microbiol. 2016;2016:2393942. DOI: 10.1155/2016/2393942
[27] Domkam IK, Sonela N, Kamgaing N, Takam PS, Gwom LC, Betilene TMA, Fokam J, Billong SC, Moukam LV, Etounou TM, Minka CSM, Ndjolo A. Prevalence and risk factors to HIV-infection amongst health care workers within public and private health facilities in Cameroon. Pan Afr Med J. 2018 Mar 19;29:158. DOI: 10.11604/pamj.2018.29.158.14073
[28] Araújo de T M, Souza de FO, Heliotério MC, Andrade de KVF, Sousa Pinho de S, Werneck GL. The high prevalence of infectious diseases among health workers indicates the need for improving surveillance. Revista Brasileira Saúde Ocupac. 2023; 48:e17. DOI: 10.1590/2317-6369/23021en2023v48e17
[29] Ganczak M, Topczewska K, Biesiada D, Korzeń M. Frequency of Occupational Bloodborne Infections and Sharps Injuries among Polish Paramedics from Selected Ambulance Stations. Int J Environ Res Public Health. 2020 Dec 23;18(1):60. DOI: 10.3390/ijerph18010060
[30] Merza MA, Mohammed SA, Qasim AM, Abdulah DM. Sero-prevalence and associated risk factors of blood-borne viral infection among healthcare workers of a tertiary referral hospital: a single-center experience. Health Probl Civiliz. 2023;17(4):328-38. DOI: 10.5114/hpc.2023.131083
[31] Altıntaş Öner B, Keskin BH, Öztürk Çerik H. Seroprevalence of hepatitis B, C and HIV infection in healthcare personnel in Turkey. J Infect Dev Ctries. 2025 Sep 30;19(9):1345-1350. DOI: 10.3855/jidc.21139
[32] Joyce MP, Kuhar DT, Brooks JT. Occupationally acquired HIV infection among health care workers—United States, 1985–2013. MMWR Morb Mortal Wkly Rep. 2015 Jan 9; 63(53):1245-9.
[33] UK Advisory Panel for Healthcare Workers Living with Bloodborne Viruses (UKAP). Integrated guidance on health clearance of healthcare workers and the management of healthcare workers living with bloodborne viruses (hepatitis B, hepatitis C and HIV). London: UK Health Security Agency; 2024 Apr. Available from: https://assets.publishing.service.gov.uk/media/677fe15cd721a08c0066560a/integrated-guidance-for-management-of-bbv-in-hcw-quick-reference-guide-April-2024-update.pdf
[34] Henderson DK, Dembry LM, Sifri CD, Palmore TN, Dellinger EP, Yokoe DS, et al; Society for Healthcare Epidemiology of America. Management of healthcare personnel living with hepatitis B, hepatitis C, or human immunodeficiency virus in US healthcare institutions. Infect Control Hosp Epidemiol. 2022 Feb;43(2):147-55. DOI: 10.1017/ice.2020.458
Attachments
| Attachment 1 | Search strategies (Attachment1_dgkh000634.pdf, application/pdf, 113.72 KBytes) |
| Attachment 2 | Quality assessment (Attachment2_dgkh000634.pdf, application/pdf, 115.71 KBytes) |




