[Randomisierte kontrollierte klinische Studie zum Einfluss der kombinierten Gabe von Rhamnose-Probiotika und oralem Nystatin auf die Besiedlung des Darms mit Candida albicans bei Neonaten mit einem Geburtsgewicht <2.500 g]
Mazyar Vakiliamini 1Reza Habibi 1
Samira Zarei 2
Roya Chegene Lorestani 3
Fatemeh Rezaeeniya 4
Farzad Mashreghi 1
Pourya Mohammadi 5
Mansour Rezaei 6
Mosayeb Rostamian 3
Hajar Motamed 7
1 Clinical Research Development Center, Imam Khomeini and Mohammad Kermanshahi and Farabi Hospitals, Kermanshah University of Medical Sciences, Kermanshah, Iran
2 Department of Microbiology, School of Sciences, Kherad Institute of Higher Education, Bushehr, Iran
3 Infectious Diseases Research Center, Health Policy and Promotion Institute, Kermanshah University of Medica Sciences, Kermanshah, Iran
4 School of Medicine, Lorestan University of Medical Sciences, Khorramabad, Iran
5 Department of Parasitology, School of Medicine, Qazvin University of Medical Sciences, Gazvin, Iran
6 Department of Biostatistics, School of Health, Kermanshah University of Medical Sciences, Kermanshah, Iran
7 Department of Pathology, School of Medicine, Kermanshah University of Medical Sciences Kermanshah, Iran
Zusammenfassung
Zielsetzung: Es sollte die Auswirkung der gleichzeitigen Einnahme von Rhamnosus-Probiotika und dem oralen Antimykotikum Nystatin auf die Hemmung der Besiedlung mit Candida (C.) albicans bei Säuglingen mit einem Gewicht <2.500 Gramm untersucht werden.
Methode: Die randomisierte, kontrollierte, klinische Studie wurde bei 104 Neonaten mit einem Geburtsgewicht <2,500 g im Mohammad Kermanshahi Hospital in Kermanshah in den Jahren 2021–22 durchgeführt. Die Neonaten wurden nach dem Zufallsprinzip in eine Interventionsgruppe (n=52) und eine Kontrollgruppe (n=52) eingeteilt. Die Kontrollgruppe erhielt viermal täglich 10 Tropfen eines Placebos oral. Die Interventionsgruppe erhielt dagegen dreimal täglich 10 Tropfen Nystatin 100.000 Einheiten oral, kombiniert mit einmal täglich Rhamnosus Lactobacillus-Tropfen oral über einen Zeitraum von 7 bis 10 d.
Ergebnisse: Am 7. Lebenstag zeigte sich in der Interventionsgruppe eine geringere Häufigkeit positiver C.-albicans-Kulturen in Stuhlproben. Darüber hinaus waren mehrere Variablen wie der Zeitpunkt des Beginns der Ernährung, die Zeit bis zum Erreichen einer Milchaufnahme von 100 ml/kg/d, die Anzahl der nil per os (NPO)-Episoden, die Gesamt-NPO-Zeit während des Krankenhausaufenthalts, die Episoden von Unverträglichkeit bis zur Entlassung und die Dauer des Krankenhausaufenthalts in der Interventionsgruppe im Vergleich zur Kontrollgruppe signifikant geringer ausgeprägt.
Diskussion: Die gleichzeitige Anwendung von Rhamnosus-Probiotika und oralem Nystatin kann bei Säuglingen mit einem Gewicht <2.500 g zu einer Verringerung der positiven Stuhlkulturergebnisse mit Nachweis von C. albicans und der Variablen Beginn der Ernährung, Zeit bis zum Erreichen einer Milchaufnahme von 100 ml/kg/d, Anzahl Tage ohne orale Nahrungsaufnahme (NPO), Gesamtdauer der NPO während des Krankenhausaufenthalts, Episoden von Unverträglichkeit bis zur Entlassung und Dauer des Krankenhausaufenthalts führen. Daher wird die gleichzeitige Verabreichung beider Mittel für Neonaten mit einem Gewicht <2.500 g empfohlen.
Schlüsselwörter
Candida albicans, enterale Besiedlung, Neonaten, Rhamnosus Probiotika, orale Nystatingabe, verkürzter Krankenhausaufenthalt, verbesserte Nahrungsaufnahme, weniger NPO Tage
Introduction
In recent years, the incidence of invasive fungal infections has notably increased in surviving low-birth-weight infants, leading to a significant mortality rate of 30% [1]. Premature and low birth weight neonates are particularly vulnerable due to their immature defense systems, delicate skin structure, fragile gastrointestinal mucosa, invasive diagnostic and treatment procedures, broad-spectrum antibiotic prescriptions, and the moist hospital environment, resulting in invasive candidiasis from intestinal colonization [2].
This colonization occurs via Candida strains transferred vertically from mothers or horizontally from healthcare workers or the hospital environment [3]. Reports indicate that 7 to 20% of premature neonates are colonized with Candida spp., predisposing them to invasive candidiasis [4] ,[5], [6], [7]. This infection intensifies with decreasing age and weight, leading to a higher prevalence of invasive fungal infections (IFIs) in very low birth weight infants (<1,500 grams) at birth, estimated at 1–9%, and in extremely low birth weight infants (<1,000 grams) at 15% [2], [8], [9], [10]. IFIs significantly impact the survival of preterm infants with prolonged hospitalization [2], [11], [12].
Moreover, early diagnosis of the infection remains challenging, resulting in treatment delays. Therefore, employing antifungal agents as a preventive measure against invasive fungal infections in this population is common. Preventing Candida ssp. intestinal colonization or treating existing cases can reduce the occurrence of necrotizing enterocolitis (NEC) or invasive candidiasis, increasing survival chances and reducing morbidity and mortality rates. Proposed methods for prophylaxis include prescribing intravenous fluconazole to infants with low birth weight or those with central venous catheters [13], the use of probiotics [14], [15], [16], the administration of oral synbiotic in low birth weight infants to prevent intestinal colonization [17], [18], and the utilization of oral antifungals such as nystatin and oral fluconazole for prophylaxis and treatment of intestinal colonization in preterm and low birth weight infants [19], [20], [21], [22], [23].
Previous investigations have addressed the individual roles of probiotic Lactobacillus (L.) rhamnosus and oral nystatin; however, the effectiveness of these two medications used in conjunction has not been assessed. Hence, this study aimed to examine the combined effect of these two drugs on preventing C. albicans intestinal colonization.
Materials and methods
This study was a comparative randomized clinical trial utilizing a triple-blind approach. In this clinical trial, the triple-blinding involved the sample groups (intervention and control), researchers, treatment teams, or any individuals involved with the samples (including statistical experts) responsible for data analysis, being unaware of the specific interventions or treatments. The study population consisted of neonates weighing <2,500 grams from June 30, 2021, to November 10, 2021, at Dr. Mohammad Kermanshahi Educational Hospital. Inclusion criteria involved birth weight <2,500 grams, hospitalization age of 72 hours or less, mothers not using oral or topical antifungals in the final months of pregnancy, and no consumption of artificial milk containing prebiotics or probiotics. Infants were excluded if they died within 7 days of birth due to congenital anomalies of the oral cavity or gastrointestinal tract, sepsis, simultaneous use of oral and injectable antifungals, or severe hemodynamic disorders.
For random allocation, a permuted randomization block method was employed to ensure balance in allocating participants into each study group. This method involved creating blocks of four (two participants in the intervention group and two in the control group). Sampling continued until the completion of the required sample size. In executing the randomization process, the individual responsible for the randomization schedule was separated from other researchers to minimize selection bias. A blinding strategy was implemented to prevent bias during and after the intervention. This entailed participants and outcome assessors being unaware of the nature of the control or intervention of groups A and B and the differences in interventions. Thus, the study was triple-blind.
According to the inclusion or exclusion criteria, 104 newborns weighing <2,500 grams were randomly assigned (for homogeneity) into two groups of 52 each. The age of these infants ranged from 1 to 3 days. The drug administration period lasted from 7 to 10 days. The control group received four doses of 10 drops of placebo (distilled water) daily. In comparison, the intervention group received three doses daily of 10 drops of oral nystatin (100,000 units) and one daily dose of 10 drops of oral L. rhamnosus, which contains 5×105 active lyophilized cells of L. rhamnosus per 10 drops. In Iran, this product is a 5-milliliter droplet stored at 2 to 8°C. No potential side effects, toxicity, or drug interactions have been reported. Two specialized C. albicans cultures were taken from each infant’s feces. The first sample was collected between 1 to 3 days of birth, before the commencement of medications, and the second was collected after 7 days. All fecal samples were promptly transferred to the laboratory and were processed within two hours of collection in a general medium, such as Sabouraud dextrose agar. For C. albicans, 0.1 gram of feces was diluted in 1.0 mL normal saline, and then 10 µL were cultured in a petri dish containing Sabouraud dextrose agar. The cultured petri dishes were incubated at 35°C for 48 hours, followed by yeast identification using germ tubes to confirm the presence of C. albicans.
The sample collection and culturing were conducted blindly, and measures were taken to prevent bias in culturing results. Following the receipt of results, comparisons were made between the infants in terms of age, gender, weight, mode of delivery, occurrence of systemic instability symptoms, NEC, intolerance, initiation of feeding, time to reach maximum feeding, frequency of nil per os (NPO) occurrences due to intolerance, duration of hospitalization, etc.
Data analysis in this study was performed using SPSS version 24 software. If necessary, the chi-squared test or Fisher’s exact test was utilized for qualitative variable comparisons. For before and after comparisons within each group, the paired t-test was used. The Kolmogorov-Smirnov (KS) test assessed normality in quantitative data. For normally distributed quantitative data, independent t-tests were employed; for non-normally distributed data, the Mann-Whitney U-test was utilized. Covariate control was implemented using analysis of covariance (ANCOVA) or categorical methods as needed for 18 confounding variables. The intention-to-treat (ITT) analysis was applied in analyzing this trial. Results were deemed statistically significant if p<0.05.
This study was registered in the Iranian Registry of Clinical Trials (IRCT registration code: IRCT 20130812014333N175). Moreover, written informed consent forms were obtained from the neonates’ parents.
The protocol was approved by the Ethics Committee of Kermanshah University of Medical Sciences (IR.KUMS.REC.1400.426).
Results
Out of 104 infants examined, in the intervention group, 26 (50%) were male and 26 (50%) were female, while in the control group, 30 (57%) were male and 22 (42%) were female.
There were no statistically significant differences between the intervention and control groups regarding gender, mode of delivery, distention, vomiting, occurrence of NEC, clinical signs, suspected and proven NEC cases, mortality within 7 days, gestational age, birth weight, weight at admission, and weight at discharge (Table 1 [Tab. 1]).
Table 1: Comparison of demographic information and clinical signs between intervention group and placebo group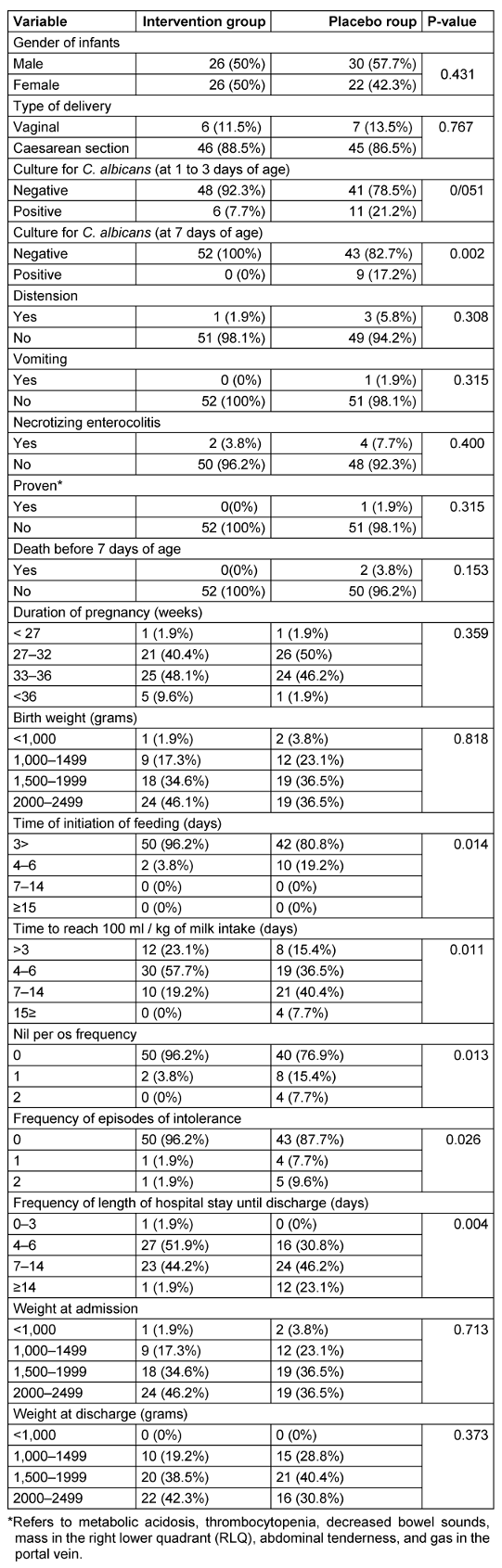
The results of culturing C. albicans from stool samples in infants aged 1 to 3 days before intervention in the intervention and control groups were 4 (7.7%) and 11 (21.2%), respectively, indicating a noticeable but not statistically significant decrease in the intervention group.
The number of positive cultures at 7 days of age in the intervention and control groups was 0 (0%) and 9 (17.3%), respectively. There was a statistically significant difference between the intervention and control groups in culturing C. albicans from stool samples at 7 days of age, indicating a significantly higher rate of positive cultures in the control group than in the intervention group (Table 1 [Tab. 1]).
The intervention and control groups significantly differed in the frequency of reaching milk intake at 100 mL/kg, with a higher percentage of children in the intervention group initiating complementary feeding before 3 days than the control group.
The frequency of zero occurrences for NPO, the total duration of NPO, and the frequency of zero occurrences for intolerance were significantly higher in the intervention group compared to the control group.
There was a statistically significant difference in the length of hospital stay until discharge (in days) between the two groups, with the intervention group showing significantly fewer days spent in the hospital until discharge than the control group.
At admission, the mean head circumference of the two groups (intervention group: 30.56±1.83, control group: 30.20±2.95) did not show a statistically significant difference. Similarly, the mean head circumference of the two groups (intervention group: 30.65±1.77, control group: 30.32±2.92) at discharge did not demonstrate a statistically significant difference (Figure 1 [Fig. 1]).
Figure 1: Mean head circumference during hospitalization and at discharge in the case and control groups
Discussion
C. albicans is a significant hospital-acquired pathogen leading to complications and mortality in premature and low birth weight infants [24]. The findings of this study revealed a substantial decrease in positive C. albicans cultures from stool samples at 7 days of age and a significant difference in mean variables, including the time until the onset of feeding, time to reach 100 mL/kg of milk intake per day, number of NPO episodes, cumulative NPO time during hospitalization, number of intolerance episodes until discharge, and length of hospital stay between the intervention and control group. However, there were no significant differences between the intervention and control groups concerning C. albicans cultures at 1 to 3 days of age, the occurrence of distension, vomiting, NEC symptoms, clinical signs, mortality within 7 days, mean age at admission, discharge weight, and length of stay at discharge.
Our study aligns with previous research in this field. Various studies have indicated that probiotics may reduce intestinal colonization in low-weight infants [16], [17], [18]. Another study reported that using L. reuteri and L. rhamnosus probiotics can prevent gastrointestinal Candida spp colonization, protect against late-onset sepsis, and reduce neurodevelopmental abnormalities in premature infants [25]. A comparative study of probiotics with nystatin demonstrated that L. reuteri is as effective as nystatin in reducing Candida colonization and might serve as a safe and effective alternative to anti-yeast drugs for low-birth-weight infants [26]. Studies have shown that probiotics can reduce Candida colonization and invasive infections due to C. albicans in rodent models [27] ,[28], [29].
Some other studies have proposed using oral anti-yeast agents such as nystatin and fluconazole for prophylaxis and treating intestinal colonization with Candida species in premature and low-weight neonates [19], [20], [21], [22]. In a randomized double-blind clinical trial, Manzoni et al. [30] reported that oral administration of L. rhamnosus and L. reuteri reduces the incidence and severity of Candida gut colonization without adverse effects. These researchers suggested that using probiotics and fluconazole to prevent fungal colonization in infants weighing <1,000 grams at birth could lead to a reduction in intestinal colonization with Candida spp.
In a systematic review conducted in 2018, 51 articles were reviewed. Among various probiotic combinations, 25 articles indicated a reduction in mortality. Seven articles confirmed a decrease in NEC occurrence. In comparison, two articles confirmed a reduction in length of stay, and three articles endorsed a decrease in the time taken for complete feeding. Provided all safety issues have been addressed, there is currently a conditional recommendation (with low certainty of evidence) to provide either L. rhamnosus GG ATCC53103 or the combination of Bifidobacterium (B.) infantis, B. lactis and Streptococcus thermophilus to reduce NEC rates [31].
Our findings also demonstrate that simultaneous use of L. rhamnosus probiotic and oral antifungal nystatin can lead to a decrease in positive C. albicans stool culture at 7 days of age and significant differences in mean variables such as the time to initiate feeding, time to reach 100 mL/kg of milk intake per day, the number of NPO episodes, cumulative NPO time during hospitalization, number of intolerance episodes until discharge, and length of hospital stay until discharge. Therefore, their usage is recommended in neonates weighing less than 2,500 grams.
Conclusions
Based on the results of the studies mentioned above, probiotics can be considered an alternative preventive and therapeutic approach. In essence, probiotics competitively exclude Candida spp., stimulate the host immune system, inhibit epithelial and mucosal adhesion, and restrain epithelial invasion [32], [33].
Notes
Competing interests
The authors declare that they have no competing interests.
Ethical approval
The protocol was approved by the Ethics Committee of Kermanshah University of Medical Sciences (IR.KUMS.REC.1400.426).
Funding
None.
Acknowledgments
We thank Dr. Mohammad Kermanshahi Hospital, Kermanshah University of Medical Sciences for their support.
Authors’ ORCIDs
- Vakiliamini M: https://orcid.org/0000-0002-9677-3846
- Habibi R: https://orcid.org/0000-0002-4579-4997
- Chegene Lorestani R: https://orcid.org/0000-0002-8137-5378
- Mohammadi P: https://orcid.org/0000-0002-4313-3255
- Rezaei M: https://orcid.org/0000-0002-6446-7289
- Rostamian M: https://orcid.org/0000-0002-1071-7019
- Motamed H: https://orcid.org/0000-0002-4419-0497
References
[1] Kelly MS, Benjamin DK Jr, Smith PB. The epidemiology and diagnosis of invasive candidiasis among premature infants. Clin Perinatol. 2015 Mar;42(1):105-17, viii-ix. DOI: 10.1016/j.clp.2014.10.008[2] Spiliopoulou A, Dimitriou G, Jelastopulu E, Giannakopoulos I, Anastassiou ED, Christofidou M. Neonatal intensive care unit candidemia: epidemiology, risk factors, outcome, and critical review of published case series. Mycopathologia. 2012 Apr;173(4):219-28. DOI: 10.1007/s11046-011-9498-3
[3] Sousa RA, Oliveira Diniz LM, Lapa Marinho FE, Gonçalves Rezende LG, Machado Carellos EM, Maia de Castro Romanelli R. Risk factors for candidemia in neonates: Systematic review and meta-analysis. J Neonatal Nurs. 2022;28(2):83-92. DOI: 10.1016/j.jnn.2021.08.
[4] Huang YC, Li CC, Lin TY, Lien RI, Chou YH, Wu JL, Hsueh C. Association of fungal colonization and invasive disease in very low birth weight infants. Pediatr Infect Dis J. 1998 Sep;17(9):819-22. DOI: 10.1097/00006454-199809000-00014
[5] Manzoni P, Farina D, Leonessa M, d'Oulx EA, Galletto P, Mostert M, Miniero R, Gomirato G. Risk factors for progression to invasive fungal infection in preterm neonates with fungal colonization. Pediatrics. 2006 Dec;118(6):2359-64. DOI: 10.1542/peds.2006-1311
[6] Singh K, Chakrabarti A, Narang A, Gopalan S. Yeast colonisation & fungaemia in preterm neonates in a tertiary care centre. Indian J Med Res. 1999 Nov;110:169-73.
[7] Mendiratta DK, Rawat V, Thamke D, Chaturvedi P, Chhabra S, Narang P. Candida colonization in preterm babies admitted to neonatal intensive care unit in the rural setting. Indian J Med Microbiol. 2006 Oct;24(4):263-7. DOI: 10.4103/0255-0857.29384
[8] Stoll BJ, Hansen N, Fanaroff AA, Wright LL, Carlo WA, Ehrenkranz RA, Lemons JA, Donovan EF, Stark AR, Tyson JE, Oh W, Bauer CR, Korones SB, Shankaran S, Laptook AR, Stevenson DK, Papile LA, Poole WK. Late-onset sepsis in very low birth weight neonates: the experience of the NICHD Neonatal Research Network. Pediatrics. 2002 Aug;110(2 Pt 1):285-91. DOI: 10.1542/peds.110.2.285
[9] Tezer H, Canpolat FE, Dilmen U. Invasive fungal infections during the neonatal period: diagnosis, treatment and prophylaxis. Expert Opin Pharmacother. 2012 Feb;13(2):193-205. DOI: 10.1517/14656566.2012.647685
[10] Ali GY, Algohary EH, Rashed KA, Almoghanum M, Khalifa AA. Prevalence of Candida colonization in preterm newborns and VLBW in neonatal intensive care unit: role of maternal colonization as a risk factor in transmission of disease. J Matern Fetal Neonatal Med. 2012 Jun;25(6):789-95. DOI: 10.3109/14767058.2011.622005
[11] Filioti J, Spiroglou K, Roilides E. Invasive candidiasis in pediatric intensive care patients: epidemiology, risk factors, management, and outcome. Intensive Care Med. 2007 Jul;33(7):1272-1283. DOI: 10.1007/s00134-007-0672-5
[12] Adams-Chapman I, Bann CM, Das A, Goldberg RN, Stoll BJ, Walsh MC, Sánchez PJ, Higgins RD, Shankaran S, Watterberg KL, Duara S, Miller NA, Heyne RJ, Peralta-Carcelen M, Goldstein RF, Steichen JJ, Bauer CR, Hintz SR, Evans PW, Acarregui MJ, Myers GJ, Vohr BR, Wilson-Costello DE, Pappas A, Vaucher YE, Ehrenkranz RA, McGowan EC, Dillard RG, Fuller J, Benjamin DK Jr; Eunice Kennedy Shriver National Institutes of Child Health and Human Development Neonatal Research Network. Neurodevelopmental outcome of extremely low birth weight infants with Candida infection. J Pediatr. 2013 Oct;163(4):961-7.e3. DOI: 10.1016/j.jpeds.2013.04.034
[13] Autmizguine J, Smith PB, Prather K, Bendel C, Natarajan G, Bidegain M, Kaufman DA, Burchfield DJ, Ross AS, Pandit P, Schell WA, Gao J, Benjamin DK Jr; Fluconazole Prophylaxis Study Team. Effect of fluconazole prophylaxis on Candida fluconazole susceptibility in premature infants. J Antimicrob Chemother. 2018 Dec;73(12):3482-3487. DOI: 10.1093/jac/dky353
[14] Matsubara VH, Wang Y, Bandara HMHN, Mayer MPA, Samaranayake LP. Probiotic lactobacilli inhibit early stages of Candida albicans biofilm development by reducing their growth, cell adhesion, and filamentation. Appl Microbiol Biotechnol. 2016 Jul;100(14):6415-6426. DOI: 10.1007/s00253-016-7527-3
[15] Hu HJ, Zhang GQ, Zhang Q, Shakya S, Li ZY. Probiotics Prevent Candida Colonization and Invasive Fungal Sepsis in Preterm Neonates: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Pediatr Neonatol. 2017 Apr;58(2):103-110. DOI: 10.1016/j.pedneo.2016.06.001
[16] Kumar S, Singhi S. Role of probiotics in prevention of Candida colonization and invasive candidiasis. J Matern Fetal Neonatal Med. 2016 Mar;29(5):818-9. DOI: 10.3109/14767058.2015.1009032
[17] Kona SK, Matlock DN. Probiotics, prebiotics, and synbiotics for preterm neonates. NeoReviews. 2018 Nov;19(11):e654-e663. DOI: 10.1542/neo.19-11-e654
[18] Vakiliamini M, Babaei H, Mohammadi M, Habibi R, Motamed H. Intestinal colonization rate of Candida albicans among low birth weight neonates after using oral synbiotic supplementation: A Randomized placebo-controlled trial. Iran J Neonatol. 2020 Sep: 11(3). DOI: 10.22038/ijn.2020.40131.1651
[19] Rundjan L, Wahyuningsih R, Oeswadi CA, Marsogi M, Purnamasari A. Oral nystatin prophylaxis to prevent systemic fungal infection in very low birth weight preterm infants: a randomized controlled trial. BMC Pediatr. 2020 Apr;20(1):170. DOI: 10.1186/s12887-020-02074-0
[20] Boselova A, Nikolinyova J, Lucanova LC, Zibolen M. Fluconazole prophylaxis of Candida infections in preterm neonates. Acta Med Martiniana. 2019;19(3):117-26. DOI: 10.2478/acm-2019-0016
[21] Leonart LP, Tonin FS, Ferreira VL, Tavares da Silva Penteado S, de Araújo Motta F, Pontarolo R. Fluconazole Doses Used for Prophylaxis of Invasive Fungal Infection in Neonatal Intensive Care Units: A Network Meta-Analysis. J Pediatr. 2017 Jun;185:129-135.e6. DOI: 10.1016/j.jpeds.2017.02.039
[22] Cabrera C, Capobiango J, Schimit T, Ferrari L, Gonzalez M, Quesada R, et al. Fluconazole for prevention of systemic infection in extremely low birth weight infants with Candidal colonization. Residência Pediátrica. 2017;6(2):80-6. DOI: 10.25060/residpediatr-2016.v6n2-08
[23] Ramasethu J. Prevention and treatment of neonatal nosocomial infections. Matern Health Neonatol Perinatol. 2017;3:5. DOI: 10.1186/s40748-017-0043-3
[24] Benjamin DK Jr, Stoll BJ, Gantz MG, Walsh MC, Sánchez PJ, Das A, Shankaran S, Higgins RD, Auten KJ, Miller NA, Walsh TJ, Laptook AR, Carlo WA, Kennedy KA, Finer NN, Duara S, Schibler K, Chapman RL, Van Meurs KP, Frantz ID 3rd, Phelps DL, Poindexter BB, Bell EF, O'Shea TM, Watterberg KL, Goldberg RN; Eunice Kennedy Shriver National Institute of Child Health and Human Development Neonatal Research Network. Neonatal candidiasis: epidemiology, risk factors, and clinical judgment. Pediatrics. 2010 Oct;126(4):e865-73. DOI: 10.1542/peds.2009-3412
[25] Romeo MG, Romeo DM, Trovato L, Oliveri S, Palermo F, Cota F, Betta P. Role of probiotics in the prevention of the enteric colonization by Candida in preterm newborns: incidence of late-onset sepsis and neurological outcome. J Perinatol. 2011 Jan;31(1):63-9. DOI: 10.1038/jp.2010.57
[26] Oncel MY, Arayici S, Sari FN, Simsek GK, Yurttutan S, Erdeve O, Saygan S, Uras N, Oguz SS, Dilmen U. Comparison of Lactobacillus reuteri and nystatin prophylaxis on Candida colonization and infection in very low birth weight infants. J Matern Fetal Neonatal Med. 2015;28(15):1790-4. DOI: 10.3109/14767058.2014.968842
[27] Leão MVP, Tavares TAA, Gonçalves E Silva CR, Dos Santos SSF, Junqueira JC, de Oliveira LD, Jorge AOC. Lactobacillus rhamnosus intake can prevent the development of Candidiasis. Clin Oral Investig. 2018 Sep;22(7):2511-2518. DOI: 10.1007/s00784-018-2347-8
[28] Villena J, Salva S, Agüero G, Alvarez S. Immunomodulatory and protective effect of probiotic Lactobacillus casei against Candida albicans infection in malnourished mice. Microbiol Immunol. 2011 Jun;55(6):434-45. DOI: 10.1111/j.1348-0421.2011.00334.x
[29] McDonough LD, Mishra AA, Tosini N, Kakade P, Penumutchu S, Liang SH, Maufrais C, Zhai B, Taur Y, Belenky P, Bennett RJ, Hohl TM, Koh AY, Ene IV. Candida albicans Isolates 529L and CHN1 Exhibit Stable Colonization of the Murine Gastrointestinal Tract. mBio. 2021 Dec;12(6):e0287821. DOI: 10.1128/mBio.02878-21
[30] Manzoni P, Mostert M, Leonessa ML, Priolo C, Farina D, Monetti C, Latino MA, Gomirato G. Oral supplementation with Lactobacillus casei subspecies rhamnosus prevents enteric colonization by Candida species in preterm neonates: a randomized study. Clin Infect Dis. 2006 Jun;42(12):1735-42. DOI: 10.1086/504324
[31] van den Akker CHP, van Goudoever JB, Szajewska H, Embleton ND, Hojsak I, Reid D, Shamir R; ESPGHAN Working Group for Probiotics, Prebiotics & Committee on Nutrition. Probiotics for Preterm Infants: A Strain-Specific Systematic Review and Network Meta-analysis. J Pediatr Gastroenterol Nutr. 2018 Jul;67(1):103-122. DOI: 10.1097/MPG.0000000000001897
[32] Ribeiro FC, Rossoni RD, de Barros PP, Santos JD, Fugisaki LRO, Leão MPV, Junqueira JC. Action mechanisms of probiotics on Candida spp. and candidiasis prevention: an update. J Appl Microbiol. 2020 Aug;129(2):175-185. DOI: 10.1111/jam.14511
[33] Rolfe RD. The role of probiotic cultures in the control of gastrointestinal health. J Nutr. 2000 Feb;130(2S Suppl):396S-402S. DOI: 10.1093/jn/130.2.396S




