[Beendigung eines Ausbruchs durch Pseudomonas aeruginosa ST111 durch Siphondesinfektion mittels zeitweiliger Absperrung des Siphons vor dem Übergang zum Abwasserabfluss]
Torsten Stein 1Martin Exner 2
Lisa-Marie Höfken 3
Katrin Schuldt 1
Axel Kramer 4
1 BDH Greifswald, Center for Neurorehabilitation, Respiratory and Intensive Care Medicine and Clinic gGmbH Paraplegia, Germany
2 Professor emeritus, Institute of Hygiene and Public Health, University of Bonn, Germany
3 National Reference Centre for multidrug-resistant gram-negative bacteria for Germany
4 Professor emeritus, Institute of Hygiene and Environmental Medicine, University Medicine Greifswald, Germany
Zusammenfassung
Einleitung: Im Ergebnis der Analyse eines Ausbruchgeschehens durch einen Carbapenem resistenten P. aeruginosa wurden die Siphons der Waschbecken in Patientenzimmern als Ursache für Kolonisationen bzw. Infektionen ermittelt. Um die Installation neuer Siphons zu vermeiden, sollte eine installatorische Lösung zur zeitweiligen Absperrung des Siphons vor dem Übergang zum Abwasserabfluss gefunden werden.
Ergebnis: Durch zeitweilige Absperrung des Ablaufs vor dem Übergang zum Abwasserabfluss mit einem Ballon gelang es, die Siphons ohne Demontage zu desinfizieren und den Ausbruch zu beenden.
Diskussion: Durch zeitweilige Absperrung des Ablaufs können Ausbrüche, ausgehend vom Siphon, beendet werden. Allerdings ist damit der Siphon nicht vor erneuter Kontamination durch den Patienten mit nachfolgender Biofilmbildung geschützt. Deshalb bleibt nur die Möglichkeit, bei bekannter Kolonisation oder Infektion des Patienten mit kritischen multiresistenten Erregern, die den Siphon kolonisieren können, den Siphon nach Entlassung des Patienten durch temporäre Absperrung zu desinfizieren. Da auch nicht resistente Erreger insbesondere bei abwehrgeschwächten Patienten Infektionen auslösen können, besteht eine Möglichkeit darin, auf Risikostationen, z.B. für Intensivtherapie, Verbrennungen, zystische Fibrose und Neonaten, auf Waschbecken zu verzichten. Grundsätzlich muss Wasser nach Patientenwaschung im Bett außerhalb des Patientenzimmers in separaten Ausgüssen entleert werden. Da durch selbstdesinfizierende Siphons die Emission aus dem Siphon deutlich reduziert wird, verbunden mit geringerer Kolonisation oder Infektion exponierter Patienten, kann deren Installation in Risikobereichen zur Reduktion nosokomialer Infektionen sinnvoll sein. In jedem Fall ist bei einem Ausbruch mit multiresistenten Erregern mit der Fähigkeit, den Siphonbereich zu kolonisieren, die Beprobung von Siphons vorzunehmen und im Fall des Nachweises die Desinfektion durch temporäre Absperrung durchzuführen.
Schlussfolgerung: Die Möglichkeit der Siphondesinfektion durch zeitweilige Absperrung ist eine technisch einfach realisierbare, ökonomische Sofortmaßnahme, bis eine Umrüstung auf Waschbecken mit rückwärtigem Abfluss oder der Einbau von Siphons mit thermischer Desinfektion vorgenommen wird.
Schlüsselwörter
Ausbruch, Carbapenem resistenter P. aeruginosa, Siphondesinfektion
Introduction
In 1991, Döring et al. [1] demonstrated that Pseudomonas (P.) aeruginosa can be transmitted from the siphon to the hands when the tap is opened due to aerosolization of the impinging water on the water standing in the siphon. More recently, siphons of both washbasins and shower drains have been repeatedly identified as a source of nosocomial transmission or outbreaks [2], [3], [4], [5].
Recurrent colonizations and infections caused by a multidrug-resistant (MDR) Pseudomonas (P.) aeruginosa strain of a carbapenemase (metallo-betalactamase) of type VIM-2 were observed over a period of 4 weeks in an intensive care unit for acute and early neurological rehabilitation. The first detection was in one patient on 20.01.2025. From April 6 to May 7 2025, the MDR P. aeruginosa strain was isolated from 9 patients. To determine the source, extensive environmental investigations were repeatedly carried out, considering the following sampling points: patient environment, beds, medical devices (perfusors, infusomats, oxygen sets), washbasins, bedpan sinks, removal area for gloves from glove boxes, disinfectant dispensers, disinfectant wipes, documentation systems, dispenser outlets, flexible nasoendoscopes, gastroscopes, drain sets and traps. As a result, the MDR P. aeruginosa strain was regularly detected in the siphons, while it was not detected at the other sampling points. Genotyping at the National Reference Center (NRZ) for Gram-negative hospital pathogens in Bochum, Germany, reconfirmed the identity of the P. aeruginosa isolates detected in the patients with the isolates from the siphons. The latter were already the initial focus as source of the outbreak, because colonizations and infections were limited to a few patient rooms.
To assess the previous situation, it should be noted that a peracetic acid-based disinfectant (concentration >1,000 ppm in the solution used) was always added to the siphon as a preventive measure after each discharge of patients with infection or colonization with carbapenem-resistant Gram-negative bacteria.
Once the cause of the outbreak had been clarified, remediation was attempted as follows in accordance with the recommendation of the Commission for Hospital Hygiene and Infection Prevention (KRINKO) at the Robert Koch Institute on hygiene requirements for wastewater-carrying systems in medical facilities [6]. The traps were replaced with new traps. After thorough mechanical cleaning, the washbasins, including the closures, were disinfected with a peracetic-acid based disinfectant. Similarly, the accessible pipe parts downstream the trap were cleaned and disinfected. Despite these measures, the new traps were recolonized with the outbreak strain within 3 days. Therefore, a solution had to be found to fill the siphon with disinfectant solution for the duration of the exposure time for the disinfectant until the transition to the wastewater pipe.
Method
The temporary blocking of the siphon before the transition to the wastewater drain was achieved with an installed balloon, which blocks the siphon to the wastewater drain for the duration of the disinfection. This option is available as a commercially available product (Geberit, International AG, Jona, Switzerland). In daily use, the installed siphon remains open to allow the water to drain off. However, to fill with the disinfectant solution, the screw-on cap at the back which allows draining is turned to the right to close it (Figure 1a [Fig. 1]).
Figure 1: Washbasin drain. a) Close the valve by turning it to the right; b) fill with disinfectant solution; c) open the valve by turning it to the left.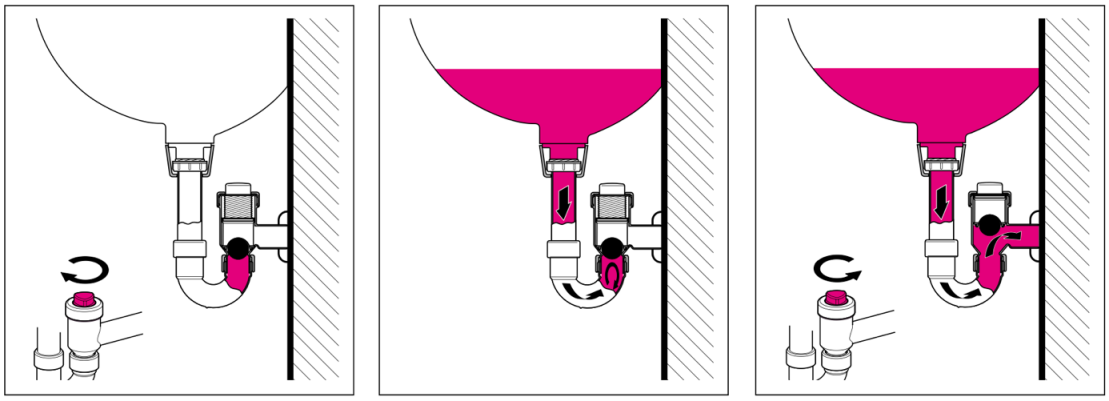
Once the disinfectant solution has been poured in (Figure 1b [Fig. 1]) and let sit for the required exposure time, the screw-on cap is turned back to the left (opened; Figure 1c [Fig. 1]) so that water drainage can resume.
Results
The temporary blocking of the siphon and disinfection succeeded in immediately stopping the outbreak. No new colonizations or infections occurred after disinfection of the blocked siphon. The outbreak strain was no longer detectable in the siphon water.
Discussion
The siphon is an open reservoir of pathogens from patients’ flora. When water enters, bacteria are ejected from the wastewater standing in the siphon over a radius of up to 1.50 m (authors’ measurement). In cases of siphon contamination of >105 colony-forming units (cfu)/mL, the transmission of bacteria to the hands of nursing staff during hand washing has been demonstrated [1]. In siphons, the genera Pseudomonas, Acidovoras, Diaphorobacter, Acinetobacter, Rhizobium, Citrobacter, Aeromonas, and Klebsiella were represented in a stable microbiome and resistome, that is, the drainome [7]. MDR P. aeruginosa, e.g., VIM-producing species, were repeatedly detected [8]. Pulmonary non-tuberculous mycobacteria (NTB) have been detected in household plumbing as possible cause of chronic rhinosinusitis [9]. Siphons have been the source of both individual infections [10] and nosocomial outbreaks. Frequently identified outbreak strains were carbapenemase-producing Enterobacteriaceae (e.g. Klebsiella pneumoniae, Escherichia coli, Enterobacter spp.), nonfermenting Gram-negatives (e.g., Pseudomonas, Burckholderia, Acinetobacter, Stenotrophomonas and Serratia spp.) [10], [11], [12], [13], [14], [15], [16], [17], [18].
The ST111 sequence type isolated here was detected during an outbreak in sink drains, two toilets, and a cleaning bucket. The outbreak containment was achieved by replacing U-bends, and cleaning buckets, and switching from quaternary ammonium compounds to oxygen-releasing disinfectant products [19]. This sequence type was the cause of another outbreak in Germany, with confirmed sink-to-patient transmission related to sink drains in two ICU rooms [20].
Due to the risk of nosocomial infections caused by siphon contamination, water-free patient care is increasingly being chosen as an alternative, at least in high-risk areas such as intensive care units and neonatal ICUs, resulting in a reduction in colonization and infections caused by Gram-negative bacteria [21], [22].
To stop an outbreak originating in the siphon, the following immediate measures were effective: cleaning the siphon 3 times a day and changing the siphon, after which an outbreak that had existed for 5 years was ended [18].
In contrast, siphon disinfection, e.g., with peracetic acid, only has a short-term effect, because the biofilm in the upper section of the drain system remained unaffected, and within two days the liquid in the P-traps was recolonized at a concentration >105 cfu/mL. After one month, this disinfecting protocol had not achieved a lasting decontamination of the sink drains [8]. A foam product containing 3.13% hydrogen peroxide and 0.05% peracetic acid resulted in significantly reduced recovery of Gram-negative bacilli on days 1, 2, and 3 after treatment, with a return to baseline by day 7 [23]. Similarly, in a longitudinal study with analysis of the drainome, elimination of critical MRE Gram-negatives was not attainable, leading to the conclusion that the optimal strategy for reduction of transmission from sinks is not known [7].
Installation of self-disinfecting sink systems (SDSS) with thermal disinfection and a biofilm-inhibiting inner surface was found to prevent pathogen reflux from the washbasin siphon trap into the washbasin. The marker organism used was a MDR P. aeruginosa. During a six-month observation period following the introduction of the self-disinfecting sink drains, no instance of nosocomial colonization of patients with a carbapenemase resistant P. aeruginosa was noted [24]. SDSS were superior to sink replacements in preventing emissions from aerosol pathogens but were unable to completely prevent the emission [25]. After replacing sink drains by SDSS the rates of microbial colonization of patients as well as the rates of incidents due to nosocomial infections were decreased [26]. Therefore, SDSS can be advantageous in special units, e.g., for cystic fibrosis patients or intensive-care neonatology.
Conclusion
The recolonization of the siphon after routine flushing of the siphon with disinfectant after each discharge of a patient who was colonized by or infected with the outbreak strain indicates that eradication of the outbreak strain from the siphon is not possible, as the strain was always detectable in subsequent sampling.
To stop the outbreak, the siphon disinfection by temporarily blocking the siphon to the wastewater is a technically simple and effective immediate measure and should be performed after each discharge of a carrier of a multidrug resistant Gram-negative bacterial species, unless the wash water is disposed of in a separate drain.
Notes
Author’s ORCID
- Kramer A: https://orcid.org/0000-0003-4193-2149
Funding
None.
Acknowledgments
The authors would like to thank Geberit International AG, Jona, Switzerland, for permission to use the figure and Mr. Karsten Nietz for installing the siphon blocking system.
Competing interests
The authors declare that they have no competing interests.
References
[1] Döring G, Ulrich M, Müller W, Bitzer J, Schmidt-Koenig L, Münst L, Grupp H, Wolz C, Stern M, Botzenhart K. Generation of Pseudomonas aeruginosa aerosols during handwashing from contaminated sink drains, transmission to hands of hospital personnel, and its prevention by use of a new heating device. Zentralbl Hyg Umweltmed. 1991 May;191(5-6):494-505.[2] Döring G, Jansen S, Noll H, Grupp H, Frank F, Botzenhart K, Magdorf K, Wahn U. Distribution and transmission of Pseudomonas aeruginosa and Burkholderia cepacia in a hospital ward. Pediatr Pulmonol. 1996 Feb;21(2):90-100. DOI: 10.1002/(SICI)1099-0496(199602)21:2<90::AID-PPUL5>3.0.CO;2-T
[3] van der Zwet WC, Nijsen IEJ, Jamin C, van Alphen LB, von Wintersdorff CJH, Demandt AMP, Savelkoul PHM. Role of the environment in transmission of Gram-negative bacteria in two consecutive outbreaks in a haematology-oncology department. Infect Prev Pract. 2022 Jun;4(2):100209. DOI: 10.1016/j.infpip.2022.100209
[4] Pieroborn C. Microbiological contamination of siphons in the neonatal intensive care unit of the Grenoble Alps university hospital and relationship with infections in premature infants. Human Health Pathol. 2023. Available from: https://dumas.ccsd.cnrs.fr/dumas-04729642
[5] Valzano F, Coda ARD, Liso A, Arena F. Multidrug-Resistant Bacteria Contaminating Plumbing Components and Sanitary Installations of Hospital Restrooms. Microorganisms. 2024 Jan;12(1):136. DOI: 10.3390/microorganisms12010136
[6] Anforderungen der Hygiene an abwasserführende Systeme in medizinischen Einrichtungen: Empfehlung der Kommission für Krankenhaushygiene und Infektionsprävention (KRINKO) beim Robert Koch-Institut. Bundesgesundheitsblatt Gesundheitsforschung Gesundheitsschutz. 2020 Apr;63(4):484-501. DOI: 10.1007/s00103-020-03118-7
[7] Snell LB, Prossomariti D, Alcolea-Medina A, Sasson M, Dibbens M, Al-Yaakoubi N, Humayun G, Charalampous T, Alder C, Ward D, Maldonado-Barrueco A, Abadioru O, Batra R, Nebbia G, Otter JA, Edgeworth JD, Goldenberg SD. The drainome: longitudinal metagenomic characterization of wastewater from hospital ward sinks to characterize the microbiome and resistome and to assess the effects of decontamination interventions. J Hosp Infect. 2024 Nov;153:55-62. DOI: 10.1016/j.jhin.2024.06.005
[8] Vanstokstraeten R, Gordts B, Verbraeken N, Blommaert L, Moretti M, De Geyter D, Wybo I. Evaluation of a peracetic-acid-based sink drain disinfectant on the intensive care unit of a tertiary care centre in Belgium. J Hosp Infect. 2024 Dec;154:45-52. DOI: 10.1016/j.jhin.2024.09.008
[9] Tichenor WS, Thurlow J, McNulty S, Brown-Elliott BA, Wallace RJ Jr, Falkinham JO 3rd. Nontuberculous Mycobacteria in household plumbing as possible cause of chronic rhinosinusitis. Emerg Infect Dis. 2012 Oct;18(10):1612-7. DOI: 10.3201/eid1810.120164
[10] Regev-Yochay G, Margalit I, Smollan G, Rapaport R, Tal I, Hanage WP, Pinas Zade N, Jaber H, Taylor BP, Che Y, Rahav G, Zimlichman E, Keller N. Sink-traps are a major source for carbapenemase-producing Enterobacteriaceae transmission. Infect Control Hosp Epidemiol. 2024 Mar;45(3):284-91. DOI: 10.1017/ice.2023.270
[11] Pitten FA, Panzig B, Schröder G, Tietze K, Kramer A. Transmission of a multiresistant Pseudomonas aeruginosa strain at a German University Hospital. J Hosp Infect. 2001 Feb;47(2):125-30. DOI: 10.1053/jhin.2000.0880
[12] Carstens A, Kepper U, Exner M, Hauri A, Kaase M, Wendt C. Plasmid-vermittelter Multispezies-Ausbruch mit Carbapenem-resistenten Enterobacteriaceae. Epid Bull. 2014;(47):455-9. Available from: https://www.rki.de/DE/Aktuelles/Publikationen/Epidemiologisches-Bulletin/2014/47_14.pdf?__blob=publicationFile&v=1
[13] Inkster T. A narrative review and update on drain-related outbreaks. J Hosp Infect. 2024 Sep;151:33-44. DOI: 10.1016/j.jhin.2024.05.016
[14] Anantharajah A, Goormaghtigh F, Nguvuyla Mantu E, Güler B, Bearzatto B, Momal A, Werion A, Hantson P, Kabamba-Mukadi B, Van Bambeke F, Rodriguez-Villalobos H, Verroken A. Long-term intensive care unit outbreak of carbapenemase-producing organisms associated with contaminated sink drains. J Hosp Infect. 2024 Jan;143:38-47. DOI: 10.1016/j.jhin.2023.10.010
[15] Lucero CA, Cohen AL, Trevino I, Rupp AH, Harris M, Forkan-Kelly S, Noble-Wang J, Jensen B, Shams A, Arduino MJ, LiPuma JJ, Gerber SI, Srinivasan A. Outbreak of Burkholderia cepacia complex among ventilated pediatric patients linked to hospital sinks. Am J Infect Control. 2011 Nov;39(9):775-8. DOI: 10.1016/j.ajic.2010.12.005
[16] Schärer V, Meier MT, Schuepbach RA, Zinkernagel AS, Boumasmoud M, Chakrakodi B, Brugger SD, Fröhlich MR, Wolfensberger A, Sax H, Kuster SP, Schreiber PW. An intensive care unit outbreak with multi-drug-resistant Pseudomonas aeruginosa - spotlight on sinks. J Hosp Infect. 2023 Sep;139:161-7. DOI: 10.1016/j.jhin.2023.06.013
[17] Volling C, Ahangari N, Bartoszko JJ, Coleman BL, Garcia-Jeldes F, Jamal AJ, Johnstone J, Kandel C, Kohler P, Maltezou HC, Maze Dit Mieusement L, McKenzie N, Mertz D, Monod A, Saeed S, Shea B, Stuart RL, Thomas S, Uleryk E, McGeer A. Are Sink Drainage Systems a Reservoir for Hospital-Acquired Gammaproteobacteria Colonization and Infection? A Systematic Review. Open Forum Infect Dis. 2021 Feb;8(2):ofaa590. DOI: 10.1093/ofid/ofaa590
[18] Lowe C, Willey B, O'Shaughnessy A, Lee W, Lum M, Pike K, Larocque C, Dedier H, Dales L, Moore C, McGeer A; Mount Sinai Hospital Infection Control Team. Outbreak of extended-spectrum β-lactamase-producing Klebsiella oxytoca infections associated with contaminated handwashing sinks(1). Emerg Infect Dis. 2012 Aug;18(8):1242-7. DOI: 10.3201/eid1808.111268
[19] Rath A, Kieninger B, Fritsch J, Caplunik-Pratsch A, Blaas S, Ochmann M, Pfeifer M, Hartl J, Holzmann T, Schneider-Brachert W. Whole-genome sequencing reveals two prolonged simultaneous outbreaks involving Pseudomonas aeruginosa high-risk strains ST111 and ST235 with resistance to quaternary ammonium compounds. J Hosp Infect. 2024 Mar;145:155-64. DOI: 10.1016/j.jhin.2024.01.009
[20] Jasuja JK, Klupp EM, Aepfelbacher M, Kampe KKW, Nentwich MF, Kluge S, Knobloch JK. Hidden transmissions of Pseudomonas aeruginosa ST111 -the importance of continuous molecular surveillance. Antimicrob Resist Infect Control. 2025 Aug;14(1):99. DOI: 10.1186/s13756-025-01619-1
[21] Inkster T, Walker J, Weinbren M. Water-free patient care: a narrative review of the literature and discussion of the pressing need for a way forward. J Hosp Infect. 2024 Oct;152:36-41. DOI: 10.1016/j.jhin.2024.06.006
[22] Low JM, Chan M, Low JL, Chua MCW, Lee JH. The impact of sink removal and other water-free interventions in intensive care units on water-borne healthcare-associated infections: a systematic review. J Hosp Infect. 2024 Aug;150:61-71. DOI: 10.1016/j.jhin.2024.05.012
[23] Jones LD, Mana TSC, Cadnum JL, Jencson AL, Silva SY, Wilson BM, Donskey CJ. Effectiveness of foam disinfectants in reducing sink-drain gram-negative bacterial colonization. Infect Control Hosp Epidemiol. 2020 Mar;41(3):280-5. DOI: 10.1017/ice.2019.325
[24] Parthé S, Welker A, Meier C, Knorr B, Schumacher S. Application study on aerosol-reducing hygienic siphons for control of a 4MRGN Pseudomonas aeruginosa outbreak on a neurological early stage rehabilitation ward. Hyg Med. 2016;41(12):E194- E199.
[25] Sissoko B, Sütterlin R, Stöber K, Schluttig A. Prevention of nosocomial infections from sink drains of washing basins. Hyg Med. 2004;29(1):20-4.
[26] Fusch C, Pogorzelski D, Main C, Meyer CL, El Helou S, Mertz D. Self-disinfecting sink drains reduce the Pseudomonas aeruginosa bioburden in a neonatal intensive care unit. Acta Paediatr. 2015 Aug;104(8):e344-9. DOI: 10.1111/apa.13005




