[Das Vestibularimplantat bei bilateraler Vestibulopathie: Eine innovative und sich stetig weiterentwickelnde Technologie]
Ying Lin 1,2Raymond van de Berg 3
Nils Guinand 4
Angelica Pérez-Fornos 4
Stan C. J. van Boxel 3
Elke M. J. Devocht 3
1 Department of Otorhinolaryngology & Head and Neck Surgery, Guangzhou Red Cross Hospital (Guangzhou Red Cross Hospital of Jinan University), Guangzhou, China
2 Institute of Otorhinolaryngology & Head and Neck Surgery, Jinan University, Guangzhou, China
3 Department of Otorhinolaryngology & Head and Neck Surgery, School for Mental Health and Neuroscience, Faculty of Health Medicine and Life Sciences, Maastricht University Medical Center, Maastricht, The Netherlands
4 Division of Otorhinolaryngology Head and Neck Surgery, Geneva University Hospitals and University of Geneva, Switzerland
Zusammenfassung
Die bilaterale Vestibulopathie (BVP) ist eine chronische vestibuläre Funktionsstörung, die durch schwerwiegende Unsicherheiten beim Gehen und durch Oszillopsie (verschwommenes Sehen bei Bewegung) gekennzeichnet ist. Aufgrund dieser Auswirkungen kann die Lebensqualität stark beeinträchtigt werden. Da vestibuläre Rehabilitationsansätze bislang nur begrenzte Erfolge zeigen, konzentrieren sich aktuelle Forschungsansätze auf die Entwicklung eines künstlichen Gleichgewichtsorgans, dem sogenannten vestibulären Implantat. Dieses Implantat bietet das Potenzial, die vestibuläre Funktion (teilweise) wiederherzustellen. Dieser Beitrag strebt an, einen umfassenden Überblick über den aktuellen Stand der Forschung zur vestibulären Implantation darzustellen. Im Fokus der Ausarbeitung stehen Kriterien zur Patientenauswahl, die Entwicklung der Implantate, die Platzierung der Elektroden, klinische Ergebnisse, Strategien zur Optimierung der Anpassung sowie zukünftige Herausforderungen.
Schlüsselwörter
Vestibularimplantation, bilaterale Vestibulopathie, Neuroprothese, Cochlea-Implantat
Introduction
Severe bilateral vestibulopathy (BVP) is a condition characterized by the loss of function in both vestibular organs, which are located in the inner ear. This dysfunction leads to the breakdown of essentials reflexes responsible for stabilizing gaze and posture. Specifically, impairment of the vestibulo-ocular reflex (VOR), the vestibulocollic reflex (VCR), and the vestibulospinal reflex (VSR) cause patients to develop blurred vision during head movements (oscillopsia) and persistent unsteadiness. These debilitating, yet often under recognized, symptoms significantly interfere with daily activities and lead to a substantial reduction in quality of life [1], [2].
A diagnosis of BVP is established based on diagnostic criteria as defined by the Bárány Society [3], [4]. These criteria encompass a combination of characteristic symptoms (unsteadiness and oscillopsia) and objective laboratory findings. The laboratory findings include: a horizontal angular VOR gain on both sides <0.6 (angular velocity 150–300°/s); and/or the sum of the maximal peak velocities of the slow phase caloric-induced nystagmus for stimulation with warm and cold water on each side <6°/s; and/or the horizontal angular VOR gain ≤0.1 upon sinusoidal stimulation on a rotatory chair (0.1 Hz, Vmax=50°/s) and/or a phase lead ≥15 degrees (time constant of ≤6 seconds).
BVP is a heterogeneous disorder with a wide range of underlying etiologies. These include ototoxicity, bilateral Menière’s disease, bilateral vestibular schwannoma, monogenetic disorders (e.g. DFNA9 and CANVAS), as well as idiopathic cases. Its prevalence is estimated to range from 28 to 81 per 100,000 adults [5], although this number is likely underestimated.
The currently recommended treatment options offered to patients with BVP consist of patient education and physiotherapy-based exercise programs [6], [7] aimed at promoting central compensation strategies. Alternative approaches have investigated sensory substitution devices, such as vibrotactile balance belts [8], or the use of noisy galvanic stimulation [9] to boost residual vestibular function. Although current clinical interventions have been reported to produce moderate effects [10] on selected measures of posture, balance and gaze stability, they cannot restore the underlying loss of rapid vestibular reflexes. As a result, in most patients with BVP, core vestibular symptoms (e.g. oscillopsia) and their markedly reduced health-related quality of life [11], generally show no clinically meaningful improvement over time [12]. Against this background, and building on the remarkable success of cochlear implants (CIs), the concept of a vestibular implant was introduced.
Evolution and current state of vestibular implant prototypes
Since the 1960s, pioneering studies have driven the development of vestibular implantation (VI). Seminal studies by Cohen and Suzuki [13] described in-vivo experiments in non-human mammals, demonstrating that nystagmus could be elicited by selective electrical stimulation of individual ampullary branches of the vestibular nerve. This study laid the conceptual foundation for the development of modern vestibular implants. The first clinical study with a multichannel VI in humans was published in 2014 [14]. It demonstrated that the vestibulo-ocular reflex could be artificially restored in humans using amplitude modulation.
Currently, a VI is still considered a research device [15], with four research groups worldwide actively investigating its application in humans [16]. Although vestibular implants (VIs) vary across different research prototypes, their overall design is similar to that of cochlear implants, consisting of an external processor and an internal receiver with an electrode array [17], [18], [19]. A VI aims to restore vestibular function either as standalone vestibular implant or in combination with a cochlear implant array. Devices may operate by the use of external motion sensors, which provide head motion input for dynamic motion-modulated stimulation, or without sensors, delivering continuous fixed pulse-train stimulation. Another principle distinction between research groups lies in the targeted implantation site of the electrode array within the vestibular end organ, namely the semicircular canals or the otolith organs.
Otolith implantation has predominantly been explored by a single research group, the Bionic Vest group (Las Palmas, Spain) [20], which is conducting a human trial involving prolonged stimulation. This non-modulating otolith-targeted implant is derived from the CI24RE cochlear implant (Cochlear Ltd), repurposed three electrodes to stimulate the saccular nerve using a constant pulse train. The system is combined with an additional cochlear electrode array. Recent results demonstrate improvements in static posture and dynamic gait resulting from continuous otolith stimulation [21].
The otolith organs encode head translation information and gravity [22] but exhibit a complex anatomical and physiological organization without clearly defined directional sensitivities [23]. In contrast, the semicircular canals encode head rotations through a relatively straightforward three-axis structure. Leveraging this defined organization, the other three research groups focus on implants targeting the semicircular canals, an approach commonly referred to as multicanal vestibular stimulation.
The University of Washington group (Seattle, USA) [19] employed a multicanal VI by Cochlear Ltd (Sydney, Australia), featuring six vestibular electrodes (two contacts per vestibular branch) and a ball reference contact. Initially designed as a vestibular-only device, the system was later combined with a 16-channel cochlear array. This implant was designed without motion sensor, reflecting its intended use as a vestibular pacemaker providing constant vestibular stimulation, for example to treat disabling Ménière’s disease. To date, this implant has only been evaluated in a limited number of patients under laboratory conditions [24].
The remaining two groups focus on restoring the vestibulo-ocular reflex using motion-modulated multicanal electrical vestibular stimulation. The Johns Hopkins group (Baltimore, USA) [25] uses a standalone semicircular canal vestibular implant with an external motion sensor. Developed in collaboration with MEDEL (Innsbruck, Austria), the system comprises a reference electrode and three electrode arrays, each containing three contacts inserted into the individual semicircular canals, allowing selection of the optimal electrode per canal. Patients have been using the device with continuous motion-modulated stimulation in daily life as part of a multi-year clinical trial with long-term follow-up [18].
The Geneva-Maastricht group investigates a combined cochleo-vestibular implant (CVI) that targets the semicircular canals [26]. This MEDEL design (Innsbruck, Austria) features three vestibular electrode branches, with one electrode implanted in each semicircular canal, alongside a cochlear electrode array containing nine contacts implanted in the cochlea. The accompanying audio-motion research processor [27] is designed to detect and process input from head movements, captured by head-mounted gyroscopes, as well as from sound, captured by microphones (Figure 1 [Fig. 1]). The most recent prototype is currently under investigation in a prospective human trial involving participants with BVP and severe sensorineural hearing loss in the implanted ear, transitioning prolonged cochlea-vestibular co-stimulation from in-hospital settings to at-home use [28].
Figure 1: Schematic representation of the investigational multicanal vestibulo-cochlear implant (VCI) as implemented by the Maastricht-Geneva research group in collaboration with the manufacturer MED-EL (Innsbruck, Austria). This figure was created in collaboration with the Swiss Foundation for Innovation and Training in Surgery (SFITS).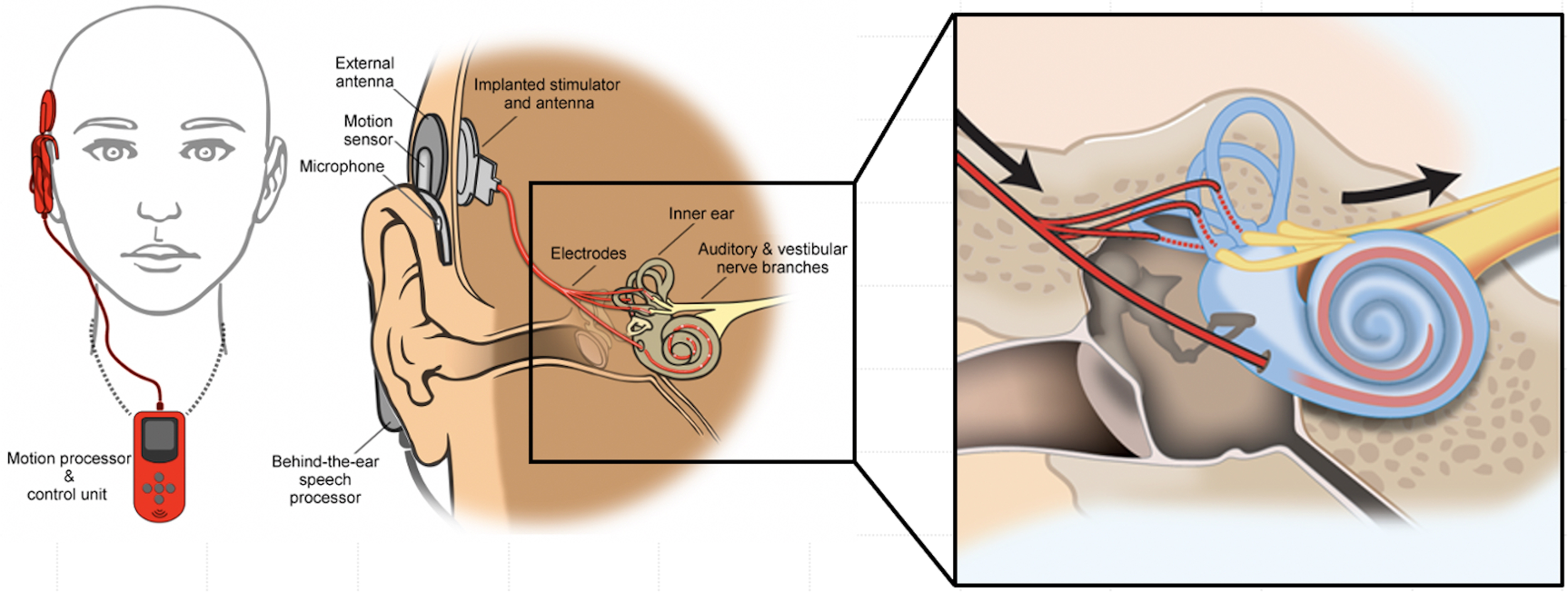
Multicanal vestibular electrode placement
The surgical procedure for vestibular implantation closely resembles that of cochlear implant surgery. However, there are two key differences: first, the vestibular system is the target organ, and second, accurate electrode placement at the intended sites is more challenging. During multicanal vestibular implantation via the intralabyrinthine approach [29], targeting the sensory epithelium of the ampullae in the three semicircular canals, this challenge is particularly significant. To improve multicanal electrode positioning and ultimately optimize response outcomes, several techniques have been investigated by the Geneva-Maastricht group. These techniques include intraoperative imaging (e.g. fluoroscopy combined with CT [30]), vestibular evoked compound action potentials [31] and intra-operative video-oculography [32]. To date, intraoperative imaging is the only method that has consistently produced reliable results. Guidance by intraoperative imaging is therefore recommended, particularly when the vestibular implant includes only one electrode per semicircular canal.
Feasibility of multicanal vestibular stimulation
Most research to date indicates that motion-modulated multicanal vestibular implantation can partially restore vestibular function by directly stimulating the vestibular nerves and bypassing the damaged sensory organs [33].
It has been shown that the VOR can be partially restored across low, middle and high-frequency head movements [14], [34]. Furthermore, the gain of electrically evoked VOR appears to be frequency dependent, similar to the gain of the ‘natural’ VOR [35]. Restoration of the VOR also resulted in a reduction of oscillopsia, reflected by improved dynamic visual acuity while walking on a treadmill [36]. This marked the first worldwide demonstration of a functional benefit achieved by vestibular implantation in humans. Additionally, otolith pathways can be activated using a semicircular canal VI, as vestibular-evoked myogenic potentials and consistent whole-body postural responses have been elicited by modulating the signal targeting the vestibular organs [37], [38]. Finally, improvements of posture, gait and quality of life have been reported after vestibular implantation, as illustrated by the research group at Johns Hopkins (USA) [18].
Taking these results into account, it can be concluded that vestibular implantation appears feasible as a clinically useful therapy in the future. Ongoing studies continue to report promising advancements in this field. Nevertheless, many challenges remain, especially regarding the fitting procedure.
Fitting a multicanal vestibular implant
The healthy vestibular system detects head movements in all directions, encompassing a broad spectrum of velocities, accelerations, and frequencies. Fitting a multicanal VI should aim to restore sensitivity across this full range, which presents the challenge of effectively encoding these complex signals via electrical stimulation. Crucially, the stimulation must be delivered in a manner that allows accurate interpretation by the central vestibular pathways where it becomes multisensory and multimodal [39].
The natural function of the semicircular canals relies on their mechanical properties. Head movements induce endolymph flow, which in turn causes deflection of the cupula. This deflection is sensed by hair cells that maintain a baseline firing rate, which is dynamically modulated in response to the degree and direction of cupula displacement. Consequently, it is hypothesized that the encoding strategy applied to vestibular stimulation should adhere to these fundamental physiological principles. Accordingly, most studies involving multicanal VIs incorporate a baseline firing rate alongside motion-driven modulation. Recent findings show that research participants rapidly acclimate to baseline vestibular stimulation with minimal discomfort [40], suggesting effective neural adaptation and supporting the safe and practical use of vestibular stimulation in daily life.
To optimize the response to multicanal vestibular stimulation, several stimulation parameters have been systematically evaluated, including pulse phase duration, stimulation rate, and pulse shape [41]. Findings indicate that higher pulse rates and longer phase durations can enhance VI performance, particularly by increasing the peak eye velocity (PEV) of the VOR. However, these enhancements are associated with trade-offs, especially in terms of reduced dynamic range and increased power consumption. Additionally, different modulation strategies, namely amplitude modulation, rate modulation and combined amplitude-rate modulation, have been compared [42]. The findings suggest that combined amplitude and rate modulation yields eye movement responses similar to those achieved with amplitude modulation alone but at a higher baseline stimulation rate. Finally, experiments [43] comparing single- and multi-canal stimulation, revealed that simultaneous multi-canal electrode activation does not simply produce the sum of single-canal responses, indicating nonlinear summation effects in the resulting eye movements. This suggests that multi-canal interactions are complex and advanced fitting strategies should account for these effects rather than relying solely on single-canal data to optimize 3D VOR restoration. Addressing this nonlinear behavior will be a key consideration in developing future implant fitting strategies, alongside evaluating approaches that compare or combine head velocity and head acceleration as inputs for VI modulation.
Future directions
Despite extensive research in this promising field, several critical aspects remain to be explored, including clinical indications, surgical techniques, and device optimization and fitting strategies.
Current indications for vestibular implantation are primarily limited to patients with BVP [15], however, emerging indications may extend to patients with Menière’s disease experiencing disabling vertigo attacks [44] or those with chronic uncompensated unilateral vestibulopathy (UVH). Although chronic unilateral vestibular hypofunction is a heterogeneous disorder, evidence shows that a subset of patients experience persistent, disabling symptoms [45] that do not respond to conventional treatment resulting in a substantially reduced quality of life [46]. For these patients, a tailored approach is warranted, in which vestibular implant could offer a future perspective, provided that careful patient selection and tailored stimulation strategies are developed. The response of patients with combined cerebellar and peripheral vestibular disorders, such as CANVAS, to vestibular stimulation remains unknown and warrants investigation in future studies. Additionally, the effect of etiology on the outcomes of vestibular stimulation is a promising area of research, as both the origin of the underlying disease and the progression timeline toward BVP (e.g., gradual onset versus recent loss) may influence implant performance. Finally, the potential benefits of bilateral implantation and the feasibility of vestibular implantation in pediatric populations warrant comprehensive future investigation [47], [48]. From an indication perspective [49], a vestibular-only implant has the potential to benefit a substantially larger proportion of patients with bilateral vestibulopathy (BVP) than a combined cochleovestibular implant, as about two-thirds of BVP patients retain residual functional hearing and are therefore not candidates for cochlear implantation [24]. For this prevalent patient group, a hearing-preserving vestibular-only approach would be more appropriate. Consequently, the primary remaining surgical concern for vestibular-only implantation is the reliable preservation of hearing [50]. While, substantial hearing loss after vestibular-only implantation has been reported in a subset of patients with preoperative residual hearing [18], [24], case reports of successful hearing preservation also exist [50]. The mechanisms underlying hearing loss after vestibular implantation and the inter-patient variability remain unclear [51], but may involve opening of the perilymphatic space and, in some cases, the endolymphatic space when the membranous labyrinth is perforated. Moreover, pressure changes during electrode insertion or postoperative factors such as local inflammation may play a role. The intralabyrinthine approach carries the highest risk, as it requires direct access to the semicircular canal ampullae. Strategies to preserve residual hearing focus on careful opening of the canals, minimizing trauma during drilling and electrode insertion, and continued refinement of electrode designs. As an alternative, extralabyrinthine [52], [53] approaches have been proposed to reduce the risk of direct labyrinthine injury. In addition, optimized intra- and post-operative radiological and audiological monitoring seems essential to detect and mitigate inner ear damage. The continued development and implementation of atraumatic (“soft surgery”) techniques is thus imperative to broaden the candidate pool for vestibular-only implantation beyond those eligible for combined cochleovestibular devices.
Regarding device optimization, an ideal processor for vestibular stimulation would be lightweight, easy to wear and operate, and have sufficient battery capacity. Incorporating multiple electrodes per vestibular lead, combined with a suitable stimulator, could enable bipolar stimulation for more localized and selective activation of the vestibular nerves, potentially reducing current spread to nontargeted ampullary nerves [54] and therefore minimizing misalignment of evoked eye responses, albeit at the cost of increased power requirements. Moreover, significant challenges remain in optimizing multicanal verstibular implant fitting [55]. Most studies suggest that smaller pulse widths [56] and pulse amplitude modulation are beneficial in humans [57]. To further enhance stimulation efficacy, it is also important to refine and individualize input-output characteristics. The input-output mapping determines how specific head movements are translated into electrical stimulus intensities delivered to the vestibular nerve, ultimately producing evoked eye movements. The goal of vestibular stimulation is to achieve precise control of eye movement responses across the full range of head orientations, velocities, and frequencies. However, both the stimulation range and the resulting VOR responses vary substantially between subjects, highlighting the need for future research on individualized fitting [18], [55], [58]. Such research should aim to optimize vestibular stimulation, elicit the most appropriate eye movement outcomes within each person’s available stimulation range, and identify the head movements most representative of daily-life situations to maximize long-term functional benefits. Finally, and perhaps most importantly, the effects of neural plasticity resulting from long-term stimulation still require thorough evaluation. After all, this plasticity may help compensate for initial mismatches between intended and actual responses, meaning that only persistent discrepancies need to be addressed and corrected through advanced fitting strategies.
Finally, it remains to be established whether vestibular implantation can evolve into a cost-effective clinical treatment. Recent health-economic analyses suggest that combined cochleovestibular implants may offer sufficient therapeutic and economic headroom [49], with cost-outcome profiles comparable to cochlear implant trajectories [59]. While these studies provide a useful reference framework for vestibular neurostimulation, they cannot yet be directly translated into reimbursement strategies. Long-term clinical outcomes, the specific benefit profile of a vestibular-only approach, as well as future device costs and surgical developments, have yet to be determined and will ultimately inform the cost-effectiveness of vestibular implantation.
Conclusion
Multicanal vestibular implantation has proven capable of partially restoring vestibular function by improving key vestibular reflexes, alleviating oscillopsia and enhancing quality of life in patients with bilateral vestibulopathy. Remaining challenges include preserving hearing and understanding the impact of etiology and neural plasticity. Future efforts should focus on refining surgical techniques, expanding indications, and advancing fitting algorithms to maximize efficacy and safety. Together, these efforts will pave the way for vestibular implantation to one day become a clinically viable therapeutic option for patients with vestibulopathy.
Notes
Acknowledgements
The authors acknowledge Johannes Seitz for his contribution in preparing the German translation of the of the manuscript’s title and abstract.
Author’s ORCID
Ying Lin: 0000-0002-6781-7010
Competing interests
The authors declare that they have no competing interests.
References
[1] Lucieer FMP, Van Hecke R, van Stiphout L, Duijn S, Perez-Fornos A, Guinand N, Van Rompaey V, Kingma H, Joore M, van de Berg R. Bilateral vestibulopathy: beyond imbalance and oscillopsia. J Neurol. 2020 Dec;267(Suppl 1):241-55. DOI: 10.1007/s00415-020-10243-5[2] Ward BK, Agrawal Y, Hoffman HJ, Carey JP, Della Santina CC. Prevalence and impact of bilateral vestibular hypofunction: results from the 2008 US National Health Interview Survey. JAMA Otolaryngol Head Neck Surg. 2013 Aug;139(8):803-10. DOI: 10.1001/jamaoto.2013.3913
[3] Strupp M, Kim JS, Murofushi T, Straumann D, Jen JC, Rosengren SM, Della Santina CC, Kingma H. Erratum to: Bilateral vestibulopathy: Diagnostic criteria Consensus document of the Classification Committee of the Bárány Society. J Vestib Res. 2023;33(1):87. DOI: 10.3233/VES-229002
[4] Strupp M, Kim JS, Murofushi T, Straumann D, Jen JC, Rosengren SM, Della Santina CC, Kingma H. Bilateral vestibulopathy: Diagnostic criteria Consensus document of the Classification Committee of the Bárány Society. J Vestib Res. 2017;27(4):177-89. DOI: 10.3233/VES-170619
[5] van Stiphout L, Szmulewicz DJ, Guinand N, Fornos AP, Van Rompaey V, van de Berg R. Bilateral vestibulopathy: a clinical update and proposed diagnostic algorithm. Front Neurol. 2023;14:1308485. DOI: 10.3389/fneur.2023.1308485
[6] Hall CD, Herdman SJ, Whitney SL, Cass SP, Clendaniel RA, Fife TD, Furman JM, Getchius TS, Goebel JA, Shepard NT, Woodhouse SN. Vestibular Rehabilitation for Peripheral Vestibular Hypofunction: An Evidence-Based Clinical Practice Guideline: FROM THE AMERICAN PHYSICAL THERAPY ASSOCIATION NEUROLOGY SECTION. J Neurol Phys Ther. 2016 Apr;40(2):124-55. DOI: 10.1097/NPT.0000000000000120
[7] Strupp M, Kern NL, Laurell G, Lehner L, Grill E, Strobl R. World-wide survey on the treatment of peripheral vestibular disorders. Front Neurol. 2025;16:1540443. DOI: 10.3389/fneur.2025.1540443
[8] Kingma H, Hougaard DD, van de Berg R. Subconscious vibrotactile stimulation improves mobility and balance in patients with bilateral vestibulopathy: adherence over 2 years. Front Neurol. 2024;15:1491195. DOI: 10.3389/fneur.2024.1491195
[9] Eder J, Kellerer S, Amberger T, Keywan A, Dlugaiczyk J, Wuehr M, Jahn K. Combining vestibular rehabilitation with noisy galvanic vestibular stimulation for treatment of bilateral vestibulopathy. J Neurol. 2022 Nov;269(11):5731-7. DOI: 10.1007/s00415-022-11033-x
[10] Porciuncula F, Johnson CC, Glickman LB. The effect of vestibular rehabilitation on adults with bilateral vestibular hypofunction: a systematic review. J Vestib Res. 2012;22(5-6):283-98. DOI: 10.3233/VES-120464
[11] Van Rompaey V, Moyaert J, Van de Heyning P, Dobbels B, van de Berg R, Guinand N, Perez-Fornos A, Mertens G. The impact of bilateral vestibulopathy on quality of life: data from the Antwerp University Hospital registry. Eur Arch Otorhinolaryngol. 2025 Sep;282(9):4537-45. DOI: 10.1007/s00405-025-09369-x
[12] Loos E, Van Stiphout L, Lucieer F, Guinand N, Pérez-Fornos A, Van Rompaey V, Desloovere C, Denys S, Verhaert N, van de Berg R. The evolution of vestibular function and health-related quality of life in bilateral vestibulopathy. Sci Rep. 2025 Mar;15(1):7476. DOI: 10.1038/s41598-025-92109-2
[13] Cohen B, Suzuki JI, Bender MB. Nystagmus Induced by Electric Stimulation of Ampullary Nerves. Acta Otolaryngol (Stockh). 1965 Jan;60(1-6):422-36. DOI: 10.3109/00016486509127026
[14] Perez Fornos A, Guinand N, van de Berg R, Stokroos R, Micera S, Kingma H, Pelizzone M, Guyot JP. Artificial balance: restoration of the vestibulo-ocular reflex in humans with a prototype vestibular neuroprosthesis. Front Neurol. 2014;5:66. DOI: 10.3389/fneur.2014.00066
[15] van de Berg R, Ramos A, van Rompaey V, Bisdorff A, Perez-Fornos A, Rubinstein JT, Phillips JO, Strupp M, Della Santina CC, Guinand N. The vestibular implant: Opinion statement on implantation criteria for research. J Vestib Res. 2020;30(3):213-23. DOI: 10.3233/VES-200701
[16] Soto E, Pliego A, Vega R. Vestibular prosthesis: from basic research to clinics. Front Integr Neurosci. 2023;17:1161860. DOI: 10.3389/fnint.2023.1161860
[17] Guyot JP, Perez Fornos A. Milestones in the development of a vestibular implant. Curr Opin Neurol. 2019 Feb;32(1):145-53. DOI: 10.1097/WCO.0000000000000639
[18] Chow MR, Ayiotis AI, Schoo DP, Gimmon Y, Lane KE, Morris BJ, Rahman MA, Valentin NS, Boutros PJ, Bowditch SP, Ward BK, Sun DQ, Treviño Guajardo C, Schubert MC, Carey JP, Della Santina CC. Posture, Gait, Quality of Life, and Hearing with a Vestibular Implant. N Engl J Med. 2021 Feb;384(6):521-32. DOI: 10.1056/NEJMoa2020457
[19] Rubinstein JT, Ling L, Nowack A, Nie K, Phillips JO. Results From a Second-Generation Vestibular Implant in Human Subjects: Diagnosis May Impact Electrical Sensitivity of Vestibular Afferents. Otol Neurotol. 2020 Jan;41(1):68-77. DOI: 10.1097/MAO.0000000000002463
[20] Ramos Macias A, Ramos de Miguel A, Rodriguez Montesdeoca I, Borkoski Barreiro S, Falcón González JC. Chronic Electrical Stimulation of the Otolith Organ: Preliminary Results in Humans with Bilateral Vestibulopathy and Sensorineural Hearing Loss. Audiol Neurootol. 2020;25(1-2):79-90. DOI: 10.1159/000503600
[21] Ramos-de-Miguel Á, Sluydts M, Falcón JC, Manrique-Huarte R, Rodriguez I, Zarowski A, Barbara M, Manrique M, Borkoski S, Lorente J, Leblanc M, Rambault A, Van Baelen E, Van Himbeeck C, Huarte A, Macías ÁR. Enhancing balance and auditory function in bilateral vestibulopathy through otolithic vestibular stimulation: insights from a pilot study on cochlea-vestibular implant efficacy. Front Neurol. 2025;16:1520554. DOI: 10.3389/fneur.2025.1520554
[22] Ramos de Miguel A, Zarowski A, Sluydts M, Ramos Macias A, Wuyts FL. The Superiority of the Otolith System. Audiol Neurootol. 2020;25(1-2):35-41. DOI: 10.1159/000504595
[23] Hageman KN, Chow MR, Roberts D, Boutros PJ, Tooker A, Lee K, Felix S, Pannu SS, Haque R, Della Santina CC. Binocular 3D otolith-ocular reflexes: responses of chinchillas to prosthetic electrical stimulation targeting the utricle and saccule. J Neurophysiol. 2020 Jan;123(1):259-76. DOI: 10.1152/jn.00883.2018
[24] Phillips JO, Ling L, Nowack A, Rebollar B, Rubinstein JT. Interactions between Auditory and Vestibular Modalities during Stimulation with a Combined Vestibular and Cochlear Prosthesis. Audiol Neurootol. 2020;25(1-2):96-108. DOI: 10.1159/000503846
[25] Della Santina CC, Migliaccio AA, Patel AH. A multichannel semicircular canal neural prosthesis using electrical stimulation to restore 3-d vestibular sensation. IEEE Trans Biomed Eng. 2007 Jun;54(6 Pt 1):1016-30. DOI: 10.1109/TBME.2007.894629
[26] van de Berg R, Stultiens JJA, van Hoof M, Van Rompaey V, Hof JR, Vermorken BL, Volpe B, Devocht EMJ, Pérez Fornos A, Postma AA, Lenoir V, Becker M, Guinand N. Vestibular Implant Surgery: How to Deal With Obstructed Semicircular Canals-A Diagnostic and Surgical Guide. J Otolaryngol Head Neck Surg. 2025;54:19160216241291809. DOI: 10.1177/19160216241291809
[27] Lanthaler D, Huebner PP, Parker MD, Griessner A, Steixner V, Zierhofer CM. A Wearable Research System for Combined Cochleo-Vestibular Stimulation. IEEE Trans Neural Syst Rehabil Eng. 2025;33:1740-50. DOI: 10.1109/TNSRE.2025.3565136
[28] Vermorken BL, Volpe B, van Boxel SCJ, Stultiens JJA, van Hoof M, Marcellis R, Loos E, van Soest A, McCrum C, Meijer K, Guinand N, Pérez Fornos A, van Rompaey V, Devocht E, van de Berg R. The VertiGO! Trial protocol: A prospective, single-center, patient-blinded study to evaluate efficacy and safety of prolonged daily stimulation with a multichannel vestibulocochlear implant prototype in bilateral vestibulopathy patients. PLoS One. 2024;19(3):e0301032. DOI: 10.1371/journal.pone.0301032
[29] van de Berg R, Guinand N, Guyot JP, Kingma H, Stokroos RJ. The modified ampullar approach for vestibular implant surgery: feasibility and its first application in a human with a long-term vestibular loss. Front Neurol. 2012;3:18. DOI: 10.3389/fneur.2012.00018
[30] Loos E, Stultiens JJA, Volpe B, Vermorken BL, Van Boxel SCJ, Devocht EMJ, van Hoof M, Postma AA, Guinand N, Pérez-Fornos A, Van Rompaey V, Denys S, Desloovere C, Verhaert N, van de Berg R. Optimizing vestibular implant electrode positioning using fluoroscopy and intraoperative CT imaging. Eur Arch Otorhinolaryngol. 2024 Jul;281(7):3433-41. DOI: 10.1007/s00405-023-08428-5
[31] van Boxel SCJ, Vermorken BL, Volpe B, Guinand N, Perez-Fornos A, Devocht EMJ, van de Berg R. Exploring applications of electrically evoked compound action potentials using the vestibulo-cochlear implant. Hear Res. 2025 Aug;464:109326. DOI: 10.1016/j.heares.2025.109326
[32] van Boxel SCJ, Vermorken BL, Volpe B, Guinand N, Perez-Fornos A, Devocht EMJ, van de Berg R. Exploring the role of the electrically evoked Vestibulo-Ocular reflex in vestibular implant surgery. Eur Arch Otorhinolaryngol. 2026 Jan;283(1):131-9. DOI: 10.1007/s00405-025-09635-y
[33] Ayiotis AI, Schoo DP, Fernandez Brillet C, Lane KE, Carey JP, Della Santina CC. Patient-Reported Outcomes After Vestibular Implantation for Bilateral Vestibular Hypofunction. JAMA Otolaryngol Head Neck Surg. 2024 Mar;150(3):240-8. DOI: 10.1001/jamaoto.2023.4475
[34] Guinand N, Van de Berg R, Cavuscens S, Ranieri M, Schneider E, Lucieer F, Kingma H, Guyot JP, Pérez Fornos A. The Video Head Impulse Test to Assess the Efficacy of Vestibular Implants in Humans. Front Neurol. 2017;8:600. DOI: 10.3389/fneur.2017.00600
[35] van de Berg R, Guinand N, Nguyen TA, Ranieri M, Cavuscens S, Guyot JP, Stokroos R, Kingma H, Perez-Fornos A. The vestibular implant: frequency-dependency of the electrically evoked vestibulo-ocular reflex in humans. Front Syst Neurosci. 2014;8:255. DOI: 10.3389/fnsys.2014.00255
[36] Starkov D, Guinand N, Lucieer F, Ranieri M, Cavuscens S, Pleshkov M, Guyot JP, Kingma H, Ramat S, Perez-Fornos A, van de Berg R. Restoring the High-Frequency Dynamic Visual Acuity with a Vestibular Implant Prototype in Humans. Audiol Neurootol. 2020;25(1-2):91-5. DOI: 10.1159/000503677
[37] Fornos AP, van de Berg R, Armand S, Cavuscens S, Ranieri M, Crétallaz C, Kingma H, Guyot JP, Guinand N. Cervical myogenic potentials and controlled postural responses elicited by a prototype vestibular implant. J Neurol. 2019 Sep;266(Suppl 1):33-41. DOI: 10.1007/s00415-019-09491-x
[38] Boutabla A, Cavuscens S, Ranieri M, Crétallaz C, Kingma H, van de Berg R, Guinand N, Pérez Fornos A. Simultaneous activation of multiple vestibular pathways upon electrical stimulation of semicircular canal afferents. J Neurol. 2020 Dec;267(Suppl 1):273-84. DOI: 10.1007/s00415-020-10120-1
[39] Angelaki DE, Cullen KE. Vestibular system: the many facets of a multimodal sense. Annu Rev Neurosci. 2008;31:125-50. DOI: 10.1146/annurev.neuro.31.060407.125555
[40] Vermorken BL, van Boxel SCJ, Volpe B, Guinand N, Pérez Fornos A, Devocht EMJ, van de Berg R. Rapid acclimatization to baseline stimulation with a multi-canal vestibulocochlear implant. Eur Arch Otorhinolaryngol. 2025 Jun;282(6):2991-3003. DOI: 10.1007/s00405-024-09184-w
[41] van Boxel SCJ, Vermorken BL, Volpe B, Guinand N, Perez-Fornos A, Devocht EMJ, van de Berg R. The vestibular implant: effects of stimulation parameters on the electrically-evoked vestibulo-ocular reflex. Front Neurol. 2024;15:1483067. DOI: 10.3389/fneur.2024.1483067
[42] van Boxel SCJ, Vermorken BL, Volpe B, Guinand N, Perez-Fornos A, Devocht EMJ, van de Berg R. Vestibular implant stimulation: pulse amplitude modulation versus combined pulse rate and amplitude modulation. J Neural Eng. 2025 Mar;22(2):026034. DOI: 10.1088/1741-2552/adc33a
[43] van Boxel SCJ, Vermorken BL, Volpe B, Guinand N, Perez-Fornos A, Devocht EMJ, van de Berg R. Combining Multi-Canal Vestibular Implant Stimulation to Restore the Vestibulo-Ocular Reflex in 3D. IEEE Trans Neural Syst Rehabil Eng. 2025;33:2449-55. DOI: 10.1109/TNSRE.2025.3579417
[44] van de Berg R, Guinand N, Ranieri M, Cavuscens S, Khoa Nguyen TA, Guyot JP, Lucieer F, Starkov D, Kingma H, van Hoof M, Perez-Fornos A. The Vestibular Implant Input Interacts with Residual Natural Function. Front Neurol. 2017;8:644. DOI: 10.3389/fneur.2017.00644
[45] Karabulut M, Van Laer L, Hallemans A, Vereeck L, Van Rompaey V, Viechtbauer W, Melliti A, van Stiphout L, Mohamad A, Pérez Fornos A, Guinand N, van de Berg R. Chronic symptoms in patients with unilateral vestibular hypofunction: systematic review and meta-analysis. Front Neurol. 2023;14:1177314. DOI: 10.3389/fneur.2023.1177314
[46] Karabulut M, Viechtbauer W, Van Laer L, Mohamad A, Van Rompaey V, Guinand N, Perez Fornos A, Gerards MC, van de Berg R. Chronic Unilateral Vestibular Hypofunction: Insights into Etiologies, Clinical Subtypes, Diagnostics and Quality of Life. J Clin Med. 2024 Sep;13(18):5381. DOI: 10.3390/jcm13185381
[47] De Kegel A, Maes L, Baetens T, Dhooge I, Van Waelvelde H. The influence of a vestibular dysfunction on the motor development of hearing-impaired children. Laryngoscope. 2012 Dec;122(12):2837-43. DOI: 10.1002/lary.23529
[48] Cushing SL, Gordon KA, Rutka JA, James AL, Papsin BC. Vestibular end-organ dysfunction in children with sensorineural hearing loss and cochlear implants: an expanded cohort and etiologic assessment. Otol Neurotol. 2013 Apr;34(3):422-8. DOI: 10.1097/MAO.0b013e31827b4ba0
[49] Lucieer FMP, Paredis S, Perez-Fornos A, Guinand N, Van Rompaey V, Kingma H, Joore M, van de Berg R. Bilateral vestibulopathy - insight in impact on quality of life and economic burden. Eur Arch Otorhinolaryngol. 2025 Oct 14;2025. DOI: 10.1007/s00405-025-09692-3
[50] van de Berg R, Lucieer F, Guinand N, van Tongeren J, George E, Guyot JP, Kingma H, van Hoof M, Temel Y, van Overbeeke J, Perez-Fornos A, Stokroos R. The Vestibular Implant: Hearing Preservation during Intralabyrinthine Electrode Insertion-A Case Report. Front Neurol. 2017;8:137. DOI: 10.3389/fneur.2017.00137
[51] Stultiens JJA, Lewis RF, Phillips JO, Boutabla A, Della Santina CC, Glueckert R, van de Berg R. The Next Challenges of Vestibular Implantation in Humans. J Assoc Res Otolaryngol. 2023 Aug;24(4):401-12. DOI: 10.1007/s10162-023-00906-1
[52] Feigl GC, Fasel JH, Anderhuber F, Ulz H, Rienmüller R, Guyot JP, Kos IM. Superior vestibular neurectomy: a novel transmeatal approach for a denervation of the superior and lateral semicircular canals. Otol Neurotol. 2009 Aug;30(5):586-91. DOI: 10.1097/MAO.0b013e3181ab9164
[53] Kos MI, Feigl G, Anderhuber F, Wall C, Fasel JH, Guyot JP. Transcanal approach to the singular nerve. Otol Neurotol. 2006 Jun;27(4):542-6. DOI: 10.1097/01.mao.0000217349.09796.d1
[54] Chiang B, Fridman GY, Dai C, Rahman MA, Della Santina CC. Design and performance of a multichannel vestibular prosthesis that restores semicircular canal sensation in rhesus monkey. IEEE Trans Neural Syst Rehabil Eng. 2011 Oct;19(5):588-98. DOI: 10.1109/TNSRE.2011.2164937
[55] Ten Hoor M, van de Berg R, Pérez Fornos A, Stultiens JJA. Electrical stimulation of the vestibular nerve: evaluating effects and potential starting points for optimization in vestibular implants. Curr Opin Otolaryngol Head Neck Surg. 2024 Oct;32(5):313-21. DOI: 10.1097/MOO.0000000000001001
[56] Grill WM Jr, Mortimer JT. The effect of stimulus pulse duration on selectivity of neural stimulation. IEEE Trans Biomed Eng. 1996 Feb;43(2):161-6. DOI: 10.1109/10.481985
[57] Nguyen TA, DiGiovanna J, Cavuscens S, Ranieri M, Guinand N, van de Berg R, Carpaneto J, Kingma H, Guyot JP, Micera S, Fornos AP. Characterization of pulse amplitude and pulse rate modulation for a human vestibular implant during acute electrical stimulation. J Neural Eng. 2016 Aug;13(4):046023. DOI: 10.1088/1741-2560/13/4/046023
[58] Guinand N, Van de Berg R, Cavuscens S, Stokroos R, Ranieri M, Pelizzone M, Kingma H, Guyot JP, Pérez Fornos A. Restoring Visual Acuity in Dynamic Conditions with a Vestibular Implant. Front Neurosci. 2016;10:577. DOI: 10.3389/fnins.2016.00577
[59] van Lieshout C, Abraham K, Smit AL, Frederix GWJ. A cost-utility analysis of cochlear implants for single sided deafness in adults and children in the Netherlands. PLoS One. 2024;19(8):e0307881. DOI: 10.1371/journal.pone.0307881




